An online shopper holds a phone in one hand, clicking "Add to Cart" and a credit card in the other hand in this photo illustration of online shopping.
Diabetes
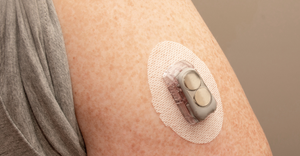
CGM: Coming Soon to Your Online Shopping CartCGM: Coming Soon to Your Online Shopping Cart
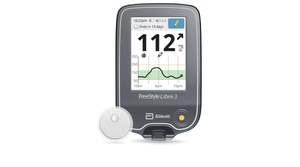
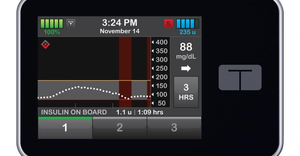
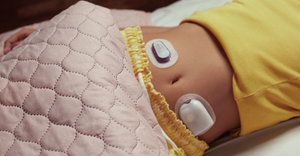
As Dexcom and Abbott both move closer to over-the-counter launches of continuous glucose monitoring sensors, analysts forecast revenue potential for the new category.
Sign up for the QMED & MD+DI Daily newsletter.






.png?width=300&auto=webp&quality=80&disable=upscale)