Challenges of Modern-Day Insulin Pump Drive Assemblies
Maxon’s Peter van Beek explores the trends and challenges in diabetic pump drive assemblies, as well as pumping systems of the future.
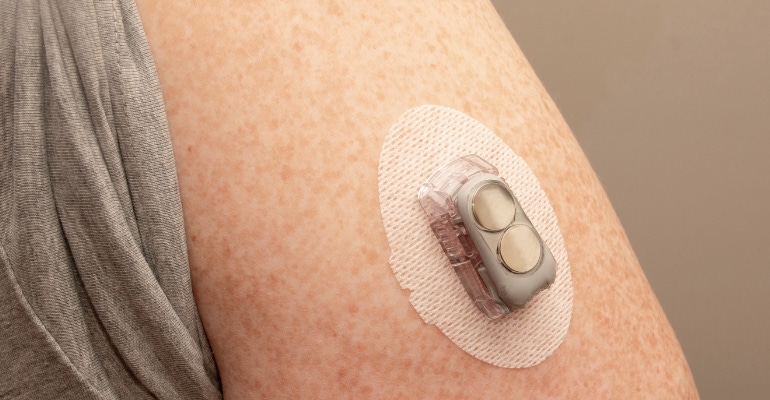
Recently, MD+DI hosted a webinar, sponsored by Maxon, exploring the challenges of modern-day insulin pump drive assemblies. The webinar, which was presented by Peter van Beek, business development manager – medical, at Maxon, focused on the history of insulin pumps, how trends in diabetic pumps have impacted drive assembly, the treatment of diabetes through different injection methods or a modular approach, and pumping systems of the future.
Van Beek noted that innovation in diabetes care has been driven by the rising rates of diabetes in the world population, creating a need for innovative solutions that allow individuals to live active and normal lives. Diabetes care that began in 1963 with Dr. Arnold Kadish’s backpack prototype that delivered insulin and glucagon is now accessible as on-body, pager-sized, self-adhesive pods that contain integrated continuous glucose monitoring (CGM) capabilities and can be controlled remotely using a cell phone.
However, with continuous innovation comes impacts to the drive assembly. Maxon understands the trend towards miniaturization and efficiency. The company achieves this efficiency essential for battery operated diabetic devices through its brushless motors, mechatronic module, and precision modular system, which combines its motor, gear, and sensor.
Looking forward, the use of CGMs, insulin pens, and on-body diabetic pumps are on the rise. But how else will diabetes be treated by pumps in the future?
Van Beek has a few ideas.
About the Author(s)
You May Also Like



.png?width=300&auto=webp&quality=80&disable=upscale)
