Diabetes Tech Integration Has Abbott and Tandem Pumped
Abbott's FreeStyle Libre 2 Plus sensor is now available for t:slim X2 insulin pump users in the United States.
January 8, 2024

Integrations help the diabetes tech world go ‘round. The latest example of this trend comes from Abbott and Tandem Diabetes Care.
The companies announced today that Tandem’s t:slim X2 insulin pump with Control-IQ technology is the first automated insulin delivery (AID) system to integrate with Abbott’s FreeStyle Libre 2 Plus, a 15-day continuous glucose monitoring (CGM) sensor. This means that, for the first time, FreeStyle Libre users in the United States are now able to experience the therapeutic benefits of a hybrid closed-loop system designed to help predict and prevent high and low blood sugar.
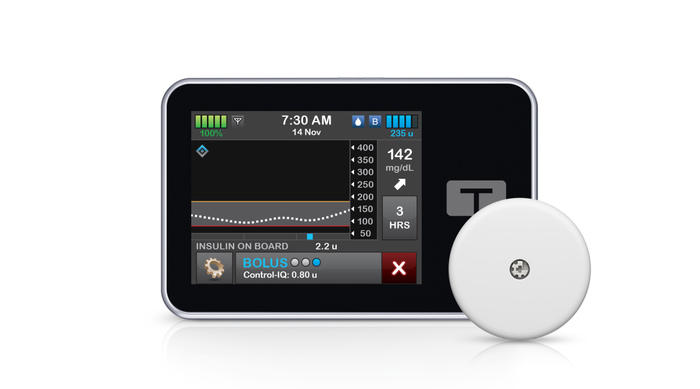
Tandem t:slim X2 insulin pump shown with Abbott's FreeStyle Libre 2 Plus continuous glucose monitoring (CGM) sensor. IMAGE COURTESY OF TANDEM DIABETES CARE
Tandem’s t:slim X2 insulin pump connects wirelessly to the FreeStyle Libre 2 Plus sensor, which sends automatic glucose readings every minute to the pump. Users can see their minute-to-minute glucose data on the pump and accompanying t:connect mobile app. The pump’s Control-IQ technology is designed to predict glucose levels 30 minutes into the future, automatically adjusting insulin delivery every five minutes based on CGM readings and can deliver automatic correction boluses (up to one per hour) to help prevent hyperglycemia.
“Tandem’s leadership in AID innovation is underscored with this milestone of launching the first insulin pump to be compatible with Abbott’s CGM technology in the U.S.,” said John Sheridan, president and CEO at Tandem. "This represents another step forward in our commitment to provide customizable solutions to help reduce burden and create new possibilities for people living with diabetes.”
The FreeStyle Libre 2 Plus is a modified version of Abbott’s FreeStyle Libre 2 sensor cleared by FDA in 2023 for use with AID systems.
Through the integration of Tandem’s insulin pump with Abbott’s CGM sensor, customers only need two sensors a month, which makes for both a more convenient and a more affordable experience, noted Jared Watkin, executive VP of Abbott’s diabetes care business.
Tandem plans to email instructions to all in-warranty t:slim X2 users in the United States offering the option to add the FreeStyle Libre 2 Plus sensor integration at no cost via a remote software update. The company said t:slim X2 pumps that have been pre-loaded with the updated software are now shipping to new customers.
About the Author(s)
You May Also Like



.png?width=300&auto=webp&quality=80&disable=upscale)
