Medical Device Regulations
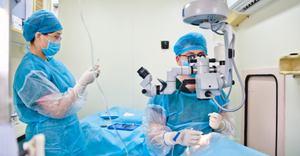
Esprit BTK System
Cardiovascular
Abbott Dissolving Stent Granted Earlier-Than-Expected FDA ApprovalAbbott Dissolving Stent Granted Earlier-Than-Expected FDA Approval
First-of-its-kind, Esprit BTK Everolimus Eluting Resorbable Scaffold System is designed to keep treat chronic limb-threatening ischemia below the knee and deliver Everolimus to support vessel healing through a stent that dissolves over time.
Sign up for the QMED & MD+DI Daily newsletter.






.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)









_illustration_with_map_of_the_European_Union_and_flag_in_the_background.png?width=300&auto=webp&quality=80&disable=upscale)



.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)



.png?width=300&auto=webp&quality=80&disable=upscale)