Medtronic's Hugo Performs Its First Robotic Surgery
The world's first Hugo patient procedure was a robotic prostatectomy performed at Clínica Santa Maria in Santiago, Chile.
June 22, 2021
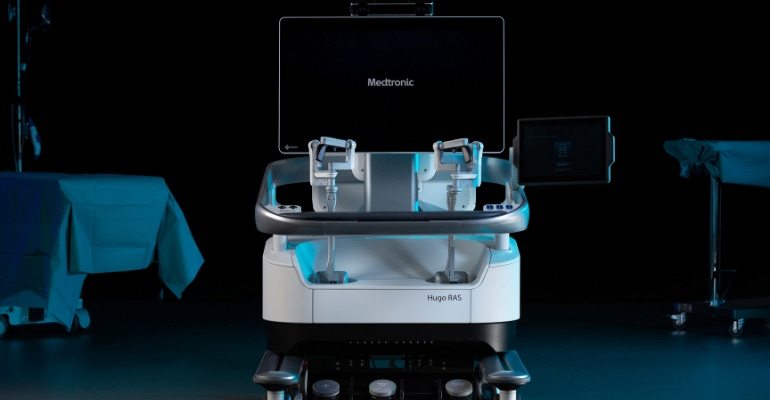
Medtronic's first soft-tissue robotic surgery is in the books.
The Dublin, Ireland-based company said the first patient procedure performed with the Hugo robotic-assisted surgery (RAS) system was a robotic prostatectomy performed June 19 by Ruben Olivares, MD, at Clínica Santa Maria in Santiago, Chile. The procedure marks the beginning of the Hugo RAS system patient registry, which will collect clinical data to support regulatory submissions around the world, Medtronic said.
A modular, multi-quadrant platform designed for a broad range of surgical procedures, the Hugo RAS system combines wristed instruments, 3D visualization, and a surgical video capture option in Touch Surgery Enterprise with dedicated support teams specializing in robotic surgery program optimization, service, and training.
"I'm proud to have performed the very first clinical procedure with the Hugo RAS system," Olivares said. "From the open console and modular platform to the instrumentation I know and trust, this innovative system is truly a partner in the OR. I feel energized by the possibility the Hugo RAS system brings to patients and clinicians and am honored to help pave the way for future surgeons, being able to offer a minimal invasive alternative on a regular basis."
In a press release, Megan Rosengarten, president of Medtronic's surgical robotics business, said that Clínica Santa Maria is the first institution to join Medtronic's Partners in Possibility program, a group of pioneering hospitals that will be among the first in the world to use the Hugo RAS system in support of the patient registry.
Clínica Santa María has more than 10 years of experience in robotic surgery, Medtronic noted.
"We are committed to providing the most advanced and innovative care to our patients. In this context, robotic surgery is a minimally invasive alternative that we have been performing with excellent results for more than a decade, with a total of more than 2,000 interventions of this type to date," said Martín Manterola, CEO at Clínica Santa María. "The alliance with Medtronic and the incorporation of the Hugo RAS system to our center places us as the first health institution in the world to use this new technology and is a milestone that reinforces the dedication and experience of our medical team."
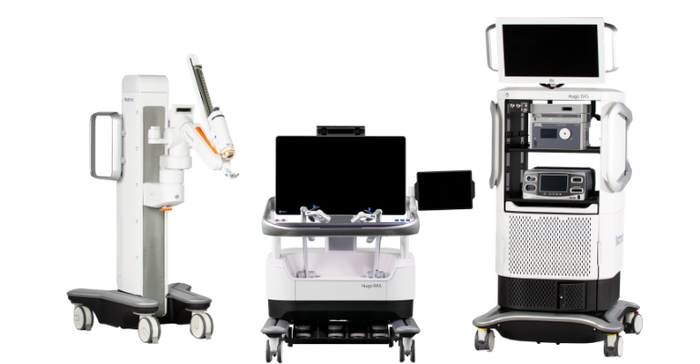
Addressing shortcomings in robotic surgery
As Medtronic approached this and other key milestones with the Hugo RAS system, MD+DI checked in March with Tracy Accardi, vice president of R&D for Medtronic's surgical robotics business, for an update on what to expect in the coming months from the company's surgical robotics team. Below is an excerpt from that conversation. The full interview can be read here.
MD+DI: What impact do you anticipate Medtronic will ultimately have in surgical robotics once the Hugo RAS system reaches the market?
Accardi: Today less than 3% of surgeries globally are being done robotically and we feel like our system could make a meaningful difference in expanding access to care to more patients in more places around the world. As our Hugo RAS system is used for the first time this year, we think it will be a great example of how we're leveraging our expertise in medtech to approach robotics in a new and meaningful way. So, we are very confident that we can make a positive impact on soft-tissue robotics.
MD+DI: What are the most significant shortcomings in robotic-assisted surgery today, and how will Medtronic's soft-tissue surgical robot address those challenges?
Accardi: Access is really key and to make that possible you really have to attack the barriers to adoption today, so we needed to provide robotic solutions that are versatile, compatible, configurable and mobile, and cost effective, as well as designing systems that are future proofed and upgradable as the technology advances rather than ones that become obsolete. So at the core of our process is a customer-centered design process and when we set out to create a new soft-tissue robotic-assisted surgery system we wanted to hear directly from surgeons and hospital executives, and we met with hundreds of them, and their input really informed our design. We heard that they wanted to be able to move the robots more easily between ORs, so we built in flexibility with a modular and upgradeable system. We also heard that they wanted compatibility with our already-trusted Medtronic surgical instrumentation. We heard that they wanted analytics that could help inform their decision making, and to confirm that the procedure was going as it needed to or to provide some insight that might not be obvious to the surgeon. Then we heard that they wanted a true partnership and we interpreted that to mean dedicated skilled engineers and system specialists to help them optimize system utilization and to provide training ... so, really those four key areas were what we heard from our surgeons and hospital executives.
About the Author(s)
You May Also Like




