Medtronic Nabs a Sweet FDA Approval in Diabetes
Medtronic also announced Tuesday afternoon that FDA has lifted the warning letter issued to its diabetes business in December 2021.
April 25, 2023
Editor's note: This story was updated April 25, 2023 to reflect important news regarding Medtronic's warning letter for the diabetes business.
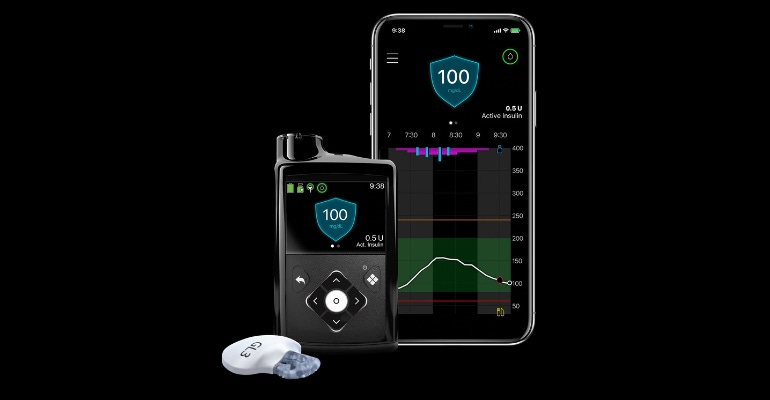
One of the Medtronic Growth Catalysts to Watch for in 2023 has been approved for sale in the United States. But the question on the minds of medtech analysts and investors is whether FDA approval of the MiniMed 780G system with the Guardian 4 sensor is enough to move the needle for the company's diabetes business.
Available in Europe since 2020, this new system is designed with meal detection technology that provides automatic adjustments and corrections to sugar levels every five minutes for both basal (background) and bolus (mealtime) insulin needs.
Medtronic originally submitted for FDA approval of the MiniMed 780G in 2021, and received a regulatory license from Health Canada in 2022. But FDA approval was delayed by quality control problems at the company's manufacturing facility, which triggered a warning letter for Medtronic's diabetes unit. That warning letter stemmed from an inspection that concluded in July 2021 related to recalls of the MiniMed 600 series insulin infusion pump, and a remote controller device for MiniMed 508 and Paradigm pumps.
"780G provides a much-needed boost for [Medtronic] at a time when other major pipeline initiatives have been challenged," Ryan Zimmerman, a medtech analyst at BTIG, noted in a report Monday.
Zimmerman said he anticipates debate amongst investors about share shift within the diabetes market, and he noted that Insulet and Tandem shares have reacted negatively on the news.
"We think the bigger question is whether this approval provides [Medtronic's] diabetes business unit with a much-needed growth boost and can it lift the entire company closer to [Medtronic's] long-term growth guidance?" Zimmerman wrote.
The analyst also pointed out that U.S. diabetes sales are about 2.7% of Medtronic's total sales, so a 100-basis point increase in U.S. diabetes growth only yields about 2 basis points of higher total Medtronic growth.
During Medtronic's fiscal third quarter earnings call in February, CEO Geoff Martha said the company's international diabetes business grew 18% on strong sales momentum of the MiniMed 780G system outside the United States. But this growth was offset by declines in the U.S. market due to the MiniMed recalls.
"We continue to invest heavily in assembling our ecosystem of durable pumps, smart pens, patch pumps, sensors, algorithms, and customer service with multiple programs under development, all with the intent of restoring strong growth of our important diabetes franchise over the coming years," Martha said.
FDA lifts warning letter for Medtronic Diabetes
Medtronic reported Tuesday that FDA lifted the warning letter received at the company's diabetes headquarters in Northridge, CA, in December 2021.
The resolution of the warning letter follows ongoing remediation actions from the company and proactive actions to continue to strengthen its quality systems, Medtronic noted. All regulatory restrictions associated with the warning letter have been resolved.
"We're very thankful to the agency for working with us so collaboratively to ensure we're able to work as quickly as possible to address the needs of the diabetes community," said Que Dallara, EVP and president of Medtronic Diabetes. "Clearing this latest regulatory milestone allows us to focus on what's most important – delivering innovations that address unmet needs for the individuals with diabetes we have the privilege to serve. It's an honor that we never take for granted and our commitment is to ensure we're putting patients first and prioritizing safety and quality above all."
Key features of the MiniMed 780G diabetes management system
According to Medtronic, the MiniMed 780G system features the lowest glucose target setting (as low as 100 mg/dL) in any automated insulin pump on the market and one that more closely mirrors the average glucose of someone not living with diabetes. With this setting, the pump will "treat to target" and will automatically deliver basal insulin adjustments and autocorrections to a set target, the company said.
It's also the only pump with an infusion set that can be worn for up to seven days, doubling wear time with advanced materials that help reduce insulin preservative loss, maintain insulin flow and stability, resulting in a reduced risk of infusion set occlusion, Medtronic noted. Combined with the Guardian 4 sensor requiring no fingersticks with SmartGuard technology, the MiniMed 780G system delivers a user-friendly design, the company said, adding that 94% of users report they're satisfied with the impact the system has on their quality of life. Importantly, users also reported remaining in SmartGuard technology 95% of the time, Medtronic noted.
"My last two years on the MiniMed 780G system as part of the clinical trial have been incredible for me," said Terry Weland, a Medtronic customer for 25 years. "The system is so easy to use. I don't know anyone who's perfect at carb counting and there are times when you're not as on top of things as you'd like to be. I don't stress over my diabetes like I used to because I know the system has my back. I love that I can sync my sensor and infusion set changes to every seven days, which streamlines my routine."
Clinical data supporting FDA approval
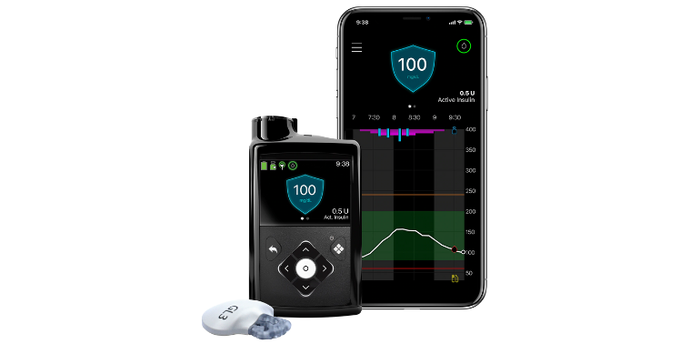
In the U.S. pivotal trial of the MiniMed 780G system, users experienced 75% time in range (blood sugar between 70-180 mg/dL), with overall time below range of 1.8%, Medtronic reported. The system provided even greater protection at night with an overnight time in range of 82%, and overnight time below range of 1.5%.
With use of the lower target of 100 mg/dL and active insulin time (AIT) set to two hours, time in range (TIR) reached 78.8% without increasing hypoglycemia. These results are mirrored by the real-world evidence across Europe where the MiniMed 780G system has been approved since 2020 and is now available in 105 countries.
Results from the randomized controlled ADAPT study evaluating the performance of the MiniMed 780G system against multiple daily injections (MDI) used in conjunction with an intermittently scanned CGM (isCGM) reinforced the significant benefits of automated insulin therapy over standard therapy. Initial six-month results, published in The Lancet Diabetes & Endocrinology, showed AHCL system users experienced a 27.6% increase in TIR and 1.4% reduction in HbA1C compared to those on MDI + isCGM without increased time in hypoglycemia. This improvement was even greater overnight with a TIR increase of 30.2%. At the close of the six-month study period, all participants on MDI + isCGM crossed over to the MiniMed 780G system. At one year, these significant improvements were reproduced in this cross-over group and sustained in those that started on AHCL therapy at the start of the trial.
The MiniMed 780G system is approved for users seven years old and above living with type 1 diabetes. Medtronic said it will begin taking pre-orders on May 15, with first shipments planned for later this summer. Customers on the company's MiniMed 770G system today will be eligible to upgrade their device to the MiniMed 780G through a no-cost, remote software upgrade.
About the Author(s)
You May Also Like




