R&D Horizons: Exploring the Internet: New Tools and Resources for Healthcare
Medical Device & Diagnostic Industry MagazineMDDI Article IndexINTO THE INTERNETOriginally Published June 2000As the Internet and related technologies provide new ways for humans to interact with machines and one another, medical device manufacturers and healthcare providers are confronting unprecedented opportunities and challenges.Gregg Nighswonger
June 1, 2000
Medical Device & Diagnostic Industry Magazine
MDDI Article Index
INTO THE INTERNET
Originally Published June 2000
Gregg Nighswonger
On June 18, 1999, ultramarathon runner Pat Farmer was preparing to begin a 15-mile leg of his run around Australia to mark the nation's Centenary of Federation. As he began the leg, viewers around the world watched as his heart rate and ECG were cybercast on the Internet in real time—providing a live demonstration of the world's first wireless Internet-based heart-monitoring system. The live cybercast was part of a research project to study the effects of extended exercise on the cardiovascular system. Electrodes applied to Farmer's chest were connected by wires to a matchbox-sized heart monitor developed by Micromedical Industries Ltd. (Labrador, Australia). A wireless data link was created by connecting the monitor to a small mobile phone carried in a pack at the runner's waist. While Farmer made his way into Brisbane, cardiac data were transmitted to the Micromedical receiving center where the heart rate was measured and ECG data were formatted for viewing on the Internet.
Commemorative marathons, such as Farmer's, may provide the stage for a dramatic display of e-health system use for monitoring and managing heart disease. It is simply one example, however, of how medical device manufacturers are taking advantage of the many opportunities offered by the Internet. Cardiology and medical imaging have been among the first medical specialties to make use of the unique capabilities offered by the Internet. But other areas of healthcare are beginning to benefit from new Internet tools and resources. Medical device makers and government regulators are exploring many of these potential applications—for design and manufacturing, research and clinical trials, regulation, teleradiology, and education.
INTERNET-BASED DESIGN ENGINEERING AND MANUFACTURING TOOLS
New resources that can support and extend the design capabilities of medical manufacturing companies are being offered by a number of e-businesses. These sites offer access to useful technical data and the services of virtual design teams, and can assist with vendor sourcing and qualification activities. Other sites provide advanced tools for remote monitoring and maintenance of manufacturing processes.
Alibre Inc. (Richardson, TX) is the first company to offer an entirely Internet-based mechanical computer-aided design service. The Internet-accessible service is available to medical device manufacturers and independent engineers and designers. According to the firm, the service "leverages the Internet as a platform to provide an integrated team environment for collaborative mechanical design and data management." The principal tool offered by the site is a 3-D mechanical design service that engineers can access like any Web page. Because the system is interactive, the software can be run from the engineer's computer
According to J. Paul Grayson, Alibre president and CEO, "Our vision is to transform the mechanical design process from one built around desktop-centric, single-user products to one based on services that are multi-user and collaborative." The company indicates that its Alibre Design is a subscription-based service that uses the concept of "collaborative engineering" to reduce required capital expenditures in order to avoid the restrictive licensing associated with traditional mechanical design software. The system is designed to provide:
Internet-enabled collaboration through real-time team modeling sessions that extend beyond viewing and markup.
Tools for establishing and managing role-based teams, collaboration sessions, and rules for sharing design data and keeping team members notified.
Simplified Internet-based data management systems to control access to design data and manage revisions.
A 3-D solid modeler for forming parts and assemblies.
A number of companies are using Internet tools to help their customers select appropriate materials, products and services. Dielectric Corp. (Menomonee Falls, WI), for example, has developed a new tool for its Web site to assist design engineers in selection of materials for the fabrication and machining of medical devices. The firm's Plastics Selector Database provides free access to pertinent technical data for product design engineering activities. The database is intended to provide the information necessary for such functions as research and development, design engineering, equipment design, and product engineering. The database allows a few known properties to be entered as a query and returns a list of materials for further review and selection. A search continues until materials are located that meet the user's criteria.
The Internet is also providing a number of new resources to assist in the control of the manufacturing process and in remote monitoring and maintenance of finished devices. Among the firms focusing on this area is eMation (Mansfield, MA), which is working to connect manufacturers to medical devices in the field via the Web. The company's embedded Internet systems enable manufacturers to perform software revisions, calibrations, and troubleshooting over the Internet—without requiring a technician on-site. According to the company, an Internet-enabled device "can even call you and warn you when a part might fail. Our technology can reduce customer downtime, which makes for better customer service." The use of Internet-embedded systems also allows real-time information exchanges between installed devices and service personnel—reducing service response times.
CLINICAL TRIALS AND RESEARCH USING THE INTERNET
The foundation of many new medical product developments—clinical trials—has become the focus of a substantial number of on-line resources. Various Web sites provide a broad range of services related to clinical studies, including information clearinghouses for potential participants, resources for clinicians, and implementation services. The Internet is being recognized as the foundation for a viable and cost-effective alternative to paper-based systems for recording, compiling, and processing clinical trials data. Use of the Internet during trials is likely to become more important as researchers face increasingly complex and expensive studies.
Developed and maintained by CenterWatch Inc. (Boston), the Web site of the Clinical Trials Listing Service has become a prominent source of information regarding studies. The site lists more than 41,000 industry- and government-sponsored clinical trials. The information provided is intended both for potential study participants and research professionals. The most recent addition to the site is a comprehensive section on studies being conducted by the National Institutes of Health, including trials from the National Cancer Institute's PDQ database. Study sponsors and clinical research organizations can use research center profiles of more than 150 clinical research centers—selecting and viewing sites by therapeutic expertise and geographic region.
ClinicalTrials.com, a site offered by Pharmaceutical Research Plus Inc. (Severna Park, MD), also provides resources for consumers, as well as healthcare professionals and researchers. The site includes clinical trials postings via a searchable database, on-line clinical trials registration for future studies, and a community outreach program intended to increase public awareness of the clinical trials process. Professional services include an investigator registry, trail recruitment assistance, and links to related services and professional organizations.
A number of medical centers and clinics, such as the Mayo Clinic (Rochester, MN), University of South Alabama College of Medicine (Mobile, AL), and the University of California at San Francisco, are also developing a Web presence focused on ongoing and planned clinical trials. These sites encompass information of current and anticipated trials, registration information, discussion groups, and searchable databases.
Nema Research Inc. (Baltimore) has focused on methods for implementing clinical trials by establishing networks of hospitals and physicians to establish eligible-patient databases. According to the firm, "Our uniqueness is in our ability to focus on both surgical and pharmaceutical trials, and through proprietary software, rapidly key in on the latest clinical research needs and match them to our available sites."
Web-based clinical trial and data collection and management systems may gradually eliminate the need for traditional paper forms that must be transcribed and processed. Phase Forward Inc. (Waltham, MA) has developed software and service systems to help clinical sponsors speed the delivery of new medical devices to market. The firm's InForm software is designed to allow clinical trials data to be accessed in real time. This allows data management and analysis to begin almost immediately after the final data have been entered, according to the firm.
Efforts to develop and assess novel medical technologies are benefiting significantly from new Internet tools and resources. Rapid electronic access to current data and information and the timely transfer of information are becoming important aspects of current research and development strategies.
Motorola Inc. (Schaumburg, IL) recently announced that it has made an equity investment in TissueInformatics Inc. (Pittsburgh), a bioinformatics firm that is creating the "world's first Virtual Tissue Bank." The project is intended to assist researchers focusing on drug discovery, tissue engineering, and evaluation of human and plant tissues, the firm states.
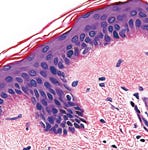 TissueInformatics' Virtual Tissue Bank will provide a database of thousands of images and related genetic information, which will be available to researchers via advanced Web-based technology.
TissueInformatics' Virtual Tissue Bank will provide a database of thousands of images and related genetic information, which will be available to researchers via advanced Web-based technology.
The multidimensional databases were devised to support the transfer of raw information into scientific knowledge by "converting advanced microscopic images of tissue into quantitative analyses of tissue structure, function, and genetic expression." The collections of both normal and diseased tissues is expected to help researchers find new treatments for a variety of diseases, including diabetes, breast cancer, and Alzheimer's disease. Researchers will be able to access the Virtual Tissue Bank through the Internet.
Says Rub Istvan, Motorola senior vice president and general manager of future businesses, "Motorola established a BioChip systems division about one year ago to develop high-quality biochip technology and products that will enable researchers to uncover the promise of genomics and, in turn, enable delivery on the dream of improved healthcare. By combining TissueInformatics' Virtual Tissue Bank data with the tissue-specific gene-expression information enabled by our biochip systems division, we can help make these databases even more useful over time. We also believe this alliance will accelerate acceptance of both the Virtual Tissue Bank and biochip technologies in practical clinical uses." TissueInformatics' first Virtual Tissue Bank is expected to be available by December 2000.
INTERNET ACCESS TO IMAGES AND DATA
The foundation of teleradiology, remote image access, and a variety of other advanced imaging capabilities has been founded on the effective use of Internet-based resources. Similarly, the Internet is offering new tools for providing remote access to patient data for both diagnostic and therapeutic applications.
Unconventional Radiology. Most radiology departments have relied on a variety of systems to generate and store reports and images. Such systems typically were not capable of communicating with one another. As a result, large, burdensome systems were needed to maintain files containing patient images and reports. A number of new Web-based systems have been developed to meet the increasing challenge of image management. Recently, ADAC Healthcare Information Systems Inc. (Houston) developed a fully integrated information- and image-management system to address these problems. ADAC's Envoi system has been designed to efficiently manage all aspects of radiology workflow and to provide a logical transition to electronic image management.
 ADAC's Web-based system for image access uses a standard browser and complies with DICOM standards.
ADAC's Web-based system for image access uses a standard browser and complies with DICOM standards.
The company indicates that "the real power of Envoi is achieved through the consolidation of what are now separate systems into one totally integrated solution. Development tools from industry leaders such as Oracle and Microsoft provide a feature-rich system that easily scales to effectively manage radiology information needs across the entire healthcare enterprise."
In addition to providing increased image access and manageability via the Internet, the sophistication of some new systems is enhancing functionality. Software being developed by Boeckeler Instruments Inc. (Tucson, AZ) is intended to offer real-time, interactive marking of images over the Internet. The firm's VIA-S70 software allows radiologists or researchers in a hospital in France, for example, to interactively view and mark a common image sent from a location in the United States.
After connecting to the Internet and using the FTP function within the software's transmittal features, each doctor can use a unique color to mark the image, add text labels, or use various pointers to communicate with the others. The markings actually exist on a separate overlay, leaving an unaltered original image. The images and overlays can be saved at any stage of discussion, and images can be printed with or without overlays, or annotated with voice and text.
Patient Monitoring. Remote access to data generated during patient monitoring is viewed as a vital aspect of effective telemedicine. The Internet is providing the basis for such capabilities. In March, Agilent Technologies Inc. (Palo Alto, CA) announced the addition of an enhancement to the Agilent Information Center. The updated system provides access to patient-monitoring information over the Internet in near real time, according to the firm. "This capability takes the core advantage of the information center—the ability to 'decentralize' patient information integrated from various patient-monitoring systems—and extends it to the Web," explains Tony Ecock, general manager of Agilent's patient-monitoring division.
Agilent's system allows clinicians to use standard internet browsers to access the information center's data. The information center combines the decision-support functions of a clinical workstation with the advantages of a central monitoring system. The system continuously displays and analyzes patient waveforms, numerics, and alarms, and allows this information to be accessed and transferred throughout the care process. Monitored information includes admission, unit-to-unit transfer, monitoring of equipment changes, and patient discharge. Ecock explains that, "Previously, these capabilities could be accessed from any workstation that was physically linked to the information center. With Web access, you don't even have to be physically linked anymore. You only need access to the Web."
NEW CHALLENGES TO REGULATION
The Internet has been viewed by many as representing a "frontier technology"—with few rules and less oversight. The free exchange of ideas and information without regulation has been considered one of the major strengths of the new medium. But it has also posed new challenges for the agencies charged with ensuring that the information being shared is accurate and truthful. For example, increasing use of the Web has revealed new avenues for promoting medical products and services. These new advertising tools, however, have also engendered more-rigorous scrutiny by FDA. (see Bottom Line column beginning on page 62)
On May 9, 2000, the agency released a warning letter issued to LaserSight Inc. (Winter Park, FL) in response to claims made on the firm's Web site regarding its LaserScan LSX excimer laser system. According to the warning letter, information on the Web site asserted that the product was approved for the treatment of myopia and hyperopia. Use of the device for hyperopia treatment, however, had not been approved, the agency stated.
The letter also noted the agency's concern about the company's claims regarding the severity of nearsightedness that is treatable with the device, and the warnings associated with that specific area of the Web site. The agency indicated that device approval requires certain basic information on intended use be included in advertising—even if the advertisement refers consumers to the product labeling.
In its response to FDA's letter, the firm explained that an unauthorized deletion of certain information from its Web site had altered its intended message. LaserSight also indicated to FDA that steps had been taken to address the problem.
Not every regulatory issue regarding Internet-based promotion is so easily resolved. Where the circulation of conventional advertising is generally contained within well-defined geographic boundaries, the Internet effectively abolishes such borders. How this will affect device regulation is still unclear. In April of this year, for example, the agency released a warning letter issued to Medtronic Inc. (Minneapolis) regarding the firm's Activa tremor control system. The letter warned that the Medtronic Web site claimed the device could treat certain problems for which it wasn't indicated. Medtronic responded that although the device was not approved in the United States for the claims made, it was indicated for those uses in other countries and that the company's Web site is international in scope.
Despite these new challenges, the Internet has proven itself to be a valuable tool to assist in device regulation. In the past 10 years, FDA has relied increasingly on the Internet to enhance the effectiveness of its activities. It has moved from a bulletin board–based system that provided simple electronic announcements of its Enforcement Report and selected documents to a sophisticated, more interactive Web site that offers a far-ranging view of its regulatory activities.
The agency is also making greater use of the Internet to support new regulatory programs. For example, CDRH is continuing pilot studies to develop an Internet-based approach to electronic registration and listing of medical device establishments with the center. Designed to facilitate annual and periodic registration updates, the system does not encompass initial registration or device listing.
CONCLUSION
From intial research, and clinical studies to use by end-users and oversight by regulators, the Internet is providing a diverse range of tools and resources for medical manufacturers and healthcare providers. The Internet is often viewed as a vast, unyielding, and barely controlled agglomeration of disorganized data. Development of new Web-based systems is often marked by a near-frantic effort to outperform competitors, secure adequate financial support, and secure the major share of a given market. The fundamental question will be whether developers and manufacturers now making this rapid-paced transition to Internet-based applications will be able to go the distance.
Gregg Nighswonger is executive editor of MD&DI.
Illustration Courtesy of TissueInformatics Inc.
Return to the MDDI June table of contents | Return to the MDDI home page
Copyright ©2000 Medical Device & Diagnostic Industry
About the Author(s)
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
