ReWalk to Demonstrate Stairs-Enabled Exoskeleton Features at Abilities Expo
Larry Jasinski, CEO and managing director of ReWalk, discusses the introduction of the stair-enabled features to the US market.
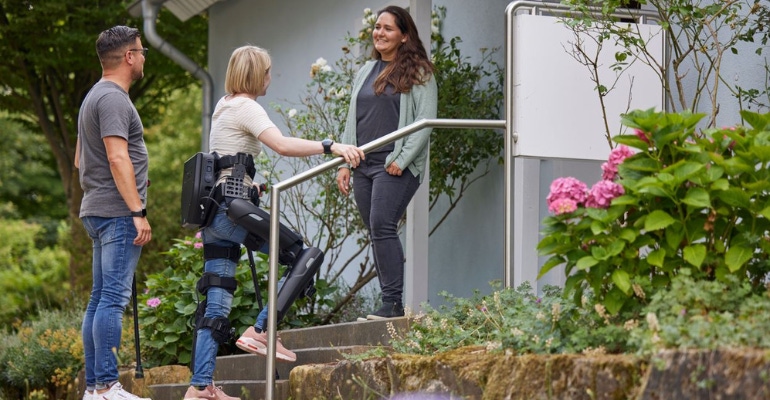
ReWalk Robotics, a provider of innovative technologies that enable mobility and wellness in rehabilitation and daily life for individuals with neurological conditions, recently announced the first United States demonstration of its stairs-enabled exoskeleton up the Abilities Expo from May 5 to 7. In light of the demonstration, MD+DI spoke with Larry Jasinski, CEO and managing director of ReWalk, to discuss the introduction of the stair-enabled features to the US market. Additionally, he highlighted who the ideal candidate is for the device, and its ease of use.
The expo is being held at the New Jersey Convention and Expo Center and is open to the public and free to attend. The demonstrations using ReWalk users will be at the company’s booth, booth #738, from 1pm and 3pm daily during the expo.
MD+DI: Who is the ideal candidate for ReWalk?
Jasinski: The ReWalk personal exoskeleton for home and community use is designed for individuals with spinal cord injury at levels T7 to L5, and who are between 5’3” and 6’2” tall and weigh less than 220 lbs. Additional indications and contraindications apply based on unique medical history and should be discussed with each patient’s prescribing physician.
MD+DI: What is the process like when a user is putting on the device? How user friendly is it to put on and use?
Jasinski: Users transfer into the device in a seated position from their wheelchair. Each device is custom configured for the individual to fit their dimensions and assistance profile, so putting on the device after it has been configured is very straightforward. The process primarily consists of securing a series of straps at the legs and waist and takes a few minutes.
During walking, users control the device through a combination of commands through the wrist-worn controller as well as subtle weight shifts to sit, stand, walk, turn, and ascend or descend stairs or curbs. Users undergo a series of training sessions in physical therapy to learn each of these skills, and each user has a designated walking companion (usually a friend, family member, etc.) who also receives training to assist the user if needed.
MD+DI: According to the ReWalk website, the ability for the ReWalk Personal Exoskeleton to ascend and descend stairs and curbs has been part of the device’s capabilities for international customers for almost a decade. What barriers were in place in the US that made the introduction of the stair walking feature difficult to approve for our market? What was the reason for the 8-year gap?
Jasinski: Yes, this feature has been available in Europe since initial CE Clearance. In order to obtain FDA clearance for this feature, real-world data from a cohort of 47 European users throughout a period of over seven years and consisting of over 18,000 stair steps was collected to demonstrate the safety and efficacy of this feature and support the FDA submission.
MD+DI: What was the US regulatory path like for the company to be able to introduce this expanded capability? Were there additional tweaks to the device to put it in compliance for US approval? If so, what were those tweaks?
Jasinski: In addition to providing real-world data from Europe to support the FDA submission, usability testing was conducted with current US ReWalk users to evaluate the usability of the new features of the ReWalk device. Some of the structural components of the legs of the exoskeleton were updated prior to this FDA submission as well, to improve the overall strength and durability of the ReWalk Exoskeleton.
MD+DI: Does Medicare and Medicaid cover the device? How about private insurance companies? Has this been a difficult process to get insurance coverage of the device?
Jasinski: ReWalk has worked extensively with the VA to ensure that a policy has been in place since 2015 to ensure that paralyzed Veterans have access to this technology. We have also worked with individual insurance companies and workers compensation to submit claims on an individual basis.
ReWalk has been working closely with the Centers for Medicare and Medicaid Services (CMS) to expand access to coverage for personal exoskeletons for Medicare Beneficiaries. Based on the feedback received to date, ReWalk Robotics is now working with individual patients and their prescribers to submit claims.
MD+DI: With the US stairs demonstration coming up in a few days, what are you most interested in the US market seeing?
Jasinski: We are excited to be able to showcase the new capabilities of the ReWalk Exoskeleton, and to highlight how members of the spinal cord injury (SCI) community can use this technology in their everyday lives to access the many benefits of walking in a variety of real-world environments. We have learned from our ReWalkers how important it is to be able to some of the things that able-bodied individuals take for granted, such as access otherwise inaccessible residential locations, use the main building entrance instead of a ramp around back, and step over stairs and curbs while out for a walk in the community. We are excited to show how adding these capabilities to the ReWalk Personal Exoskeleton will increase the opportunities for individuals to use their exoskeletons in new ways and new places in order to maximize the benefits of walking.
MD+DI: What is the company most excited about now that the stair-walking feature is available in the US?
Jasinski: We are excited to see how users can take advantage of these new features to increase their opportunities to incorporate the health benefits of walking into their everyday lives. This is a major milestone for individuals in the SCI community to improve access to real world environments and to expand the use of personal exoskeletons.
MD+DI: Does ReWalk have any competitors in the space that include a stairs feature?
Jasinski: No, ReWalk is the only personal exoskeleton that has a stairs feature. In fact, the device received “Breakthrough Device” designation from the FDA due to being the first and only device of its kind to expand ambulatory access for paralyzed individuals to everyday environments that include stairs or curbs.
About the Author(s)
You May Also Like



.png?width=300&auto=webp&quality=80&disable=upscale)
