The company is recalling thousands of embolization devices because there is a risk of the delivery system's wires and tubes fracturing and breaking off during placement, retrieval, or movement of the device.
September 20, 2021
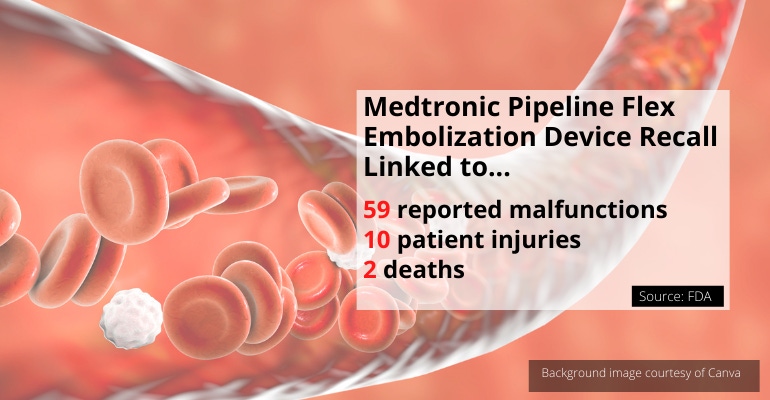
At least two people have died, and 10 others have been seriously injured after a procedure to treat brain aneurysms when a piece of a medical device made by Medtronic fractured and broke off inside their bloodstream.
Medtronic is recalling its Pipeline Flex Embolization Device and Pipeline Flex Embolization Device with Shield Technology because there is a risk that the delivery system's wires and tubes will fracture and break off during placement, retrieval, or movement of the device. There have been 59 reported device malfunctions associated with the recall, which Medtronic initiated in July. The recall includes 8,825 devices that were distributed between April 18, 2019 and August 13, 2020.
The recalled devices are permanent mesh cylinders (stents) braided from platinum and tungsten and cobalt-chromium-nickel alloy wires. These devices are intended for the treatment of brain aneurysms that bulge or balloon out the sides of the blood vessel. The devices include a guidewire-based delivery system used to place the implant inside the patient.
Medtronic inherited these devices through its $49.9 billion Covidien deal in early 2015, and Covidien had previously acquired the devices when it bought ev3 in 2010 for $2.6 billion. The subsidiary's offical name is Micro Therapeutics but it is doing business as ev3 Neurovascular.
According to the FDA notice issued Monday, fractured pieces of these devices could be left inside the patient's brain bloodstream. It is also possible that attempts to retrieve the fractured pieces may make the patient’s condition worse, the agency noted. The fragments can also cause other serious adverse health consequences such as continued blockage of blood vessels, stroke, and death.
This represents the ninth Medtronic recall that FDA has issued a notice about this year. Four of those recalls were related to the now-discontinued HeartWare HVAD system. Other Medtronic recalls this year include the recall of seven of its implantable cardioverter defibrillators (ICDs) and cardiac resynchronization therapy devices (CRT-Ds) due to the risk of a shortened battery life; and a recall of the Valiant Navion thoracic stent graft systems due to patient safety issues observed in a global cinical trial. FDA has issued a total of 45 recall notices so far this year.
About the Author(s)
You May Also Like




