Research Defines User and Customer Needs
October 1, 1997
R&D and Design Play Important Role in Development of New Biopsy Forceps
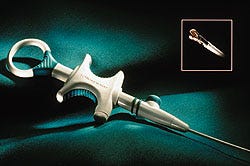
Developing a medical device is challenging enough when the product category has been pioneered by others. However, being the first to market with an innovative product is fraught with risks equal to the potential benefits.
G I Supply, a division of ChekMed Systems Inc. (Camp Hill, PA), faced this daunting task when it began developing the Respose forceps, the industry's first "resposable" biopsy forceps, which combines a reusable handle with a disposable tip. G I Supply discerned a market for the product within the fast-growing group of disposable forceps users.
G I Supply faced two key challenges. The company realized that not only would the product have to meet the needs of very particular users, but also that a strong product identity and a well orchestrated introduction would be essential to achieve instant recognition. G I Supply hired BOLT, an R&D and design firm based in Charlotte, NC, to help it meet these challenges.
Research Defines User and Customer Needs
To better understand the potential market, the design team launched a two-pronged research effort, blending user-needs research with market research to create a clear picture of both the customer and the user. User-needs information was acquired through user-task analyses, interviews with clinicians, and observation and videotaping of biopsy procedures. Market research was focused on the people who make purchasing decisions, and data were acquired through qualitative focus groups and interviews with clinicians, hospital department managers, and administrators. Through these research efforts, a model of the end-user and customer needs began to emerge.
The dimensions of this model relating to the end-user included the need for a kink-resistant coil spring to enhance the reliability of the reusable handle as compared with other products whose coil spring often became kinked during use; an ergonomic handle design; user-oriented features including various jaw styles, cautery capability, and a flush port to enhance cleaning; the degree of ease and security that must be designed into the tip attachment method (the tip incorporates the spike and jaws which actually cut the tissue); the ease-of-use and safety issues that must be addressed in the product design and discussed in the product literature; and the preferred reprocessing methods.
The customer-related facets of the model included the price points at which the product was perceived as being either too expensive compared to competitors' products or too inexpensive to be of appropriate quality; the preferred number of disposable tips to be sold within each package; the required clinical references and reprocessing information for the reusable handle; the potential barriers to sales; and the advantage of a unique handle appearance that could set the product apart from the competition.
Design Difficulties
Armed with this user/customer model, BOLT began an intensive design and engineering effort. The mechanical attachment of the tip was developed for ease of use and reliability. The handle was designed to be attractive, comfortable, and easy to use. The production materials were researched, tested, and specified to meet the rigors of use and reprocessing. Solid-modeling CAD software was used to develop models for illustrations and for stereolithography rapid prototypes. These renderings and rapid prototypes were shown to a clinical-users group for evaluation and input, and ultimately the CAD database was used to produce the final tooling for the injection-molded handle components.
The tip attachment method presented a particularly thorny design challenge. Research had shown that this attachment must be quick and easy, inherently safe, and clearly perceived as such prior to purchase. G I Supply had partnered with Marlow Surgical Technologies (Willoughby, OH), owners of a patent for disposable tips on medical devices, to manufacture the Respose. However, while Marlow's products were all laparoscopic devices, the biopsy forceps is a much smaller instrument, and therefore the tip attachment method used by Marlow would not be acceptable to users of the Respose.
After weeks of design, prototyping, and testing, the design team developed a tip attachment method that uses the properties of nickel-titanium alloy (nitinol) to provide a snap feature that allows the tip to be easily attached and removed. The team also developed a transparent tip cover that gives the user a handle to grasp while attaching a new tip, and also gives protection during removal of a used tip.
Creating the Right Identity
While the designers and engineers were detailing the product, the BOLT identity specialists were developing a strategy with G I Supply for the product's introduction. Positioning the product against the competition was the focus of the product-identity program.
Referring to the market research, BOLT developed an identity statement that defined the graphic identity of the product. Similar to a corporate mission statement, the identity statement succinctly describes the appropriate messages to be communicated through the brand identity. Once the statement was approved, it became the foundation for BOLT's development of the Respose name, product logo, product packaging, and sales literature. BOLT also helped G I Supply to design its trade show booth for the rollout of the new product.
Frank Carter, CEO of ChekMed, recalls, "It was important that we present a cohesive image to our market with the introduction of this unique device. The product design, the engineering detail, the packaging, the sales collateral, even the booth had to reflect our corporate character and our commitment to our customers."
Successful product development requires more than just a good designit requires a comprehensive team approach to determine the product's identity and a well-planned strategy for its introduction. Clearly, the Respose met all of these requirements.
To learn more about R&D and design services from BOLT, call 704/372-2658.
MPMN is actively seeking success stories like this. If your company has one to tell, please contact Editor Ursula Jones at 3340 Ocean Park Blvd., Ste. 1000, Santa Monica, CA 90405; 310/392-5509.
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
