Representation Matters in Clinical Trials
Medtronic and Boston Scientific share the efforts they have taken to ensure diverse representation in clinical trials.
February 13, 2023
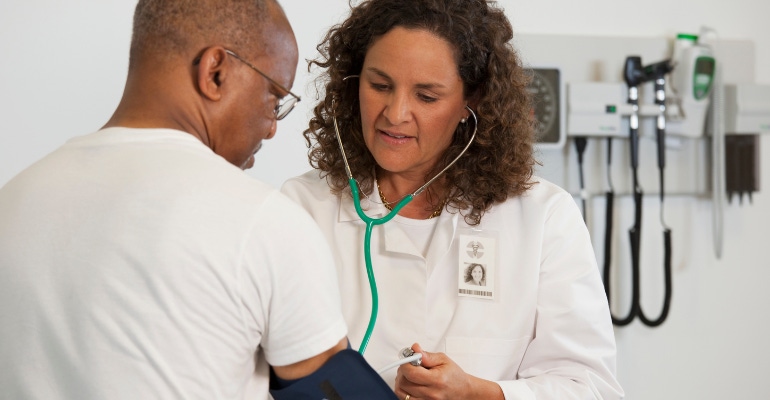
Clinical trials are an early step in device and therapy development which have significant impact on the types of products available to patients. The demographics of patients in these trials carries an impact on products, making diversity in clinical trials key to ensure all patient needs are being evaluated, and targeted for treatments. Medtronic and Boston Scientific share the efforts the companies have undertaken to broaden the patients in their clinical trials.
“We cannot provide the best therapies across the world unless we fully represent the range of people who are affected,” said Laura Mauri, MD, senior vice president, chief scientific, medical, and regulatory officer at Medtronic. “Diverse representation in our research and development is not only a social imperative that is needed to interrupt the cycle of health inequity, but also a clinical necessity to fully understand our products. Diversity allows us to better reflect the entirety of the population we’re trying to serve, improve device efficacy across that entire population, and ultimately improve outcomes for a broader group of patients and communities.”
Seeking to broaden access to clinical trials
Medtronic has actively sought to increase the diversity of patients in its trials, primarily by working with physicians to reach patients and outreach through varied methods. “We’re improving our ability to track representation across our hundreds of studies and assess what is working to reduce gaps between research and technology and the people living with the diseases we treat,” Mauri said. “We’re being mindful of bias, especially unintended or unconscious bias in research. We are engaging with physicians from diverse backgrounds and communities and looking beyond traditional research institutions to extend the breadth of communities included, co-developing our efforts with local healthcare facilities. COVID accelerated digital transformation, which is also allowing us to reach patients directly through social media.”
The company has seen success as a result of its outreach efforts, especially in areas where patients have not been as likely to utilize device options.
“More than 34 million people live with diabetes, however, Black and Hispanic Americans with diabetes are two to three times less likely to use medical technology available to treat the disease compared with white Americans,” Mauri said. “Medtronic is working to remove some of the barriers that lead to this disparity, partnering with diabetes treatment centers to provide tools to clinicians to increase diverse representation in studies and to provide relevant education to patients.”
A pilot study has demonstrated a 24% increase among racial and ethnic minority patients.
Reducing inequalities in TAVR options
Further, Medtronic has initiated a program to reach more patients with aortic stenosis who could be helped with transcatheter aortic valve replacement (TAVR).
“People of color are offered this minimally invasive option at significantly lower rates, according to research,” Mauri said. “Medtronic has launched a pilot program to better understand and address these disparities. We collaborated with several health systems to explore the root causes and are launching programs to address treatment barriers in African American and Hispanic and Latino patients.”
Medtronic has also completed enrolment for a trial comparing TAVR treatments and diverse population including women and patients of color.
Boston Scientific has also committed to inclusion in its clinical research, an effort that was part of its 20-year program to increase access to research, and which has evolved over the years.
“Our Close the Gap program started nearly two decades ago to organize and concentrate our efforts to reach women and people of color who so often suffer disproportionately higher rates of, and worse outcomes from, a range of diseases our devices and therapies treat,” said Camille Chang Gilmore, vice president of human resources and chief diversity, equity, and inclusion officer at Boston Scientific. “While we started with initiatives that largely focused on awareness and screenings — participating in health fairs, for example — we have learned over the years that as a medtech company, we have the opportunity to play a more focused role. That includes developing resources for healthcare providers to understand disease presentation and more effectively engage with women and patients of color; convening conversations between physicians and those traditionally underserved patient populations; and advancing clinical trial diversity.”
Increased access to trials
Boston Scientific's success with the program was noted in the recruitment and results of a recent trial.
“In 2016 we reported out on the PLATINUM Diversity study, the first of its kind to study how contemporary stent technology works in diverse populations and can help improve care for diverse people. In addition to traditional clinical measures, we looked at social and economic information to gain more holistic insights about barriers to better outcomes,” said Chang Gilmore. “The work continues, and subsets of that data are still revealing new insights today.”
Coming out of that study, Boston Scientific took away many learnings about recruitment efforts to reach a broader population.
“In the process, we dispelled a number of myths about the difficulty of diverse trial recruitment and actually ran ahead of schedule,” said Chang Gilmore. “A key learning, which sounds obvious today, was the importance of site selection and engaging diverse investigators, which in turn underscores the importance of deepening our relationships with diverse physicians and equipping all physicians with the resources to connect with diverse patients.”
Recruitment of diverse patients in clinical trials often hinges on working with communities in which patients receive care and have relationships with a provider.
In Boston Scientific’s efforts to eliminate health inequity and reach all patient populations, Michael Jaff, MD, vice president of clinical affairs, innovation and technology and chief medical officer for Boston Scientific's peripheral interventions business, shared: “What we’ve learned is that patients have the most trust in their own healthcare provider, and that by engaging more of the providers who serve underrepresented groups in clinical research, we can make the most meaningful progress. Increasing awareness of the opportunities and benefits for these communities is a vital piece of the puzzle, and we see it working.”
PAD research through community outreach
“One recent example is our ELEGANCE registry ... studying the impact of our advanced drug-eluting treatments for leg artery blockage, also known as [peripheral arterial disease, or PAD],” Jaff said. “Historically, women and patients in underrepresented minorities have been infrequently included in clinical trials of novel treatments for PAD. We made a deliberate decision to commit all efforts to enroll a minimum of 40% of the population as women and 40% as underrepresented minorities.”
The trial has surpassed that marker with efforts in communities where patients are currently undergoing care.
“We’ve been working in close collaboration with local hospitals and patient advocacy groups to expand our network of investigators and engage those clinicians who have trusted relationships in their communities but may not have previously been involved in medical research,” Jaff said.
About the Author(s)
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
