Common Mistakes in Medical Sealer Equipment Designs
December 10, 2010
|
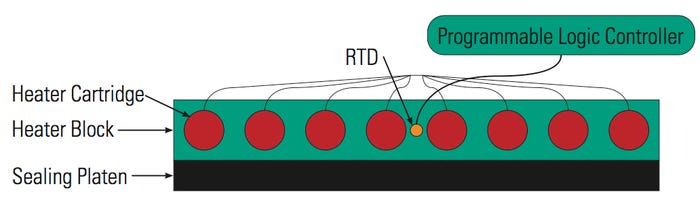
Figure 1. [Click to enlarge] A typical setup for controlling temperature in the platen. |
Based on ISO 11607, medical devices delivered in a sterile state should be designed, manufactured, and packed to ensure that they are sterile, under documented storage and transportation conditions, until the barrier system is damaged or opened.1 There should be a documented process validation program demonstrating the efficacy and reproducibility of all sterilization and packaging processes. Along with the sterilization process, some of the packaging operations that can affect sterile barrier system integrity are forming, sealing, capping (or some other closure system), cutting, and process handling.
To be compliant with ISO 11607 part 2, medical device companies must have a validation program to demonstrate repeatability and reproducibility of the sealing process. There are some common mistakes that can occur during the process of developing a packaging sealing process.
As FDA continues to regulate the processes involved in medical sterile packaging systems, the need for reducing variation on these processes becomes more critical. Variability and noises can cause a company’s process to drift or be out of control, even when sealing process parameters are not changed. But there are ways to improve the process.
Equipment Documentation
As part of the equipment design process, a user requirement specification (URS) is written to establish every requirement relating to product safety, identity, strength, productivity, and quality, among other elements. As part of the URS, documented equipment tolerances are established for the three major critical process parameters involved in a sealing process: temperature, pressure, and dwell time.
After both parties have approved the URS, the equipment supplier develops hardware design (HDS) and software design specifications (SDS). In this phase, the supplier provides documentation of how the process will function. Once these documents are discussed with the end-user, equipment failure modes and effects analysis (FMEA) must be conducted. From this analysis, equipment operational challenges are developed and source code is finally programmed to mitigate the failure modes generated by this analysis. This step is sometimes forgotten by medical device companies. Instead, the equipment operational challenges are established based on experience with no evidence of where these challenges came from.
FMEA helps the design phase by identifying the critical aspects of the process and determining ways of monitoring and controlling them in the event that a certain scenario occurs. This analysis determines the operational challenges as well as at which points the equipment should be tested to ensure that the sealer conforms to the requirements listed in the URS, HDS, and SDS.
Parameter tolerances are also part of the URS. For example, the document might say that the sealing platen or bar shall be ±3° from the set point. But what is most important is seeing how the equipment manufacturer will achieve this requirement.
Sealers can be hardwired or PLC driven depending on the model and the controls needed for the type of application. There has been a tendency to use sealers that are PLC driven because they provide greater flexibility in terms of process or design changes without causing major modifications to the machine. Hardwired machines typically have a separate temperature controller to manage the temperature while PLC-driven machines can control the temperature with the process integral device feature embedded in the PLC.
Temperature, dwell time, and pressure are typical factors involved in a sealing process. Let’s see how each factor can be addressed as part of the equipment design to reduce variation.
Temperature
Both custom and standard sealer equipment can be used to seal sterile packaging systems, depending on the application requirements. There are various methods of controlling the temperature in the platen. The most common platform is using a heating block and a platen that is bolted or otherwise attached to the block as presented in Figure 1.
This system has the resistance temperature detector (RTD) mounted in the heat source or what is called the heating block. The heating block is warmed up and heat is transferred to the sealing platen by conduction. Aluminum blocks are typically used, but other materials can also be used depending on the temperature or speed at which the process is developed.
Most systems come with one RTD to monitor the temperature across the heating block. It is usually mounted in the middle of the block. However, depending on the size of the heating block, one RTD may not provide adequate temperature control.
When the RTD is mounted in the heating block, temperature variation is precisely controlled because the RTD is very close to the heating elements. But what really happens with this design? Consider the following:
The temperature at which the package is sealed is far from the point at which the temperature is read.
There will be a temperature differential between the readout and the real temperature at which packages are sealed.
It takes more time to detect and react to temperature changes across the sealing platen surface.
The heatwave can be different depending on the temperature conditions where the sealer is placed, which may cause the surface temperature to change even though the sealer is displaying a steady temperature.
There is no way to detect a surface temperature problem in the event that the platen becomes loose or does not have the same thermal transfer as when it was originally installed.
Temperature distribution across the platen is taken for granted because it is being monitored in only one place. Any temperature variation within the platen will not be detected.
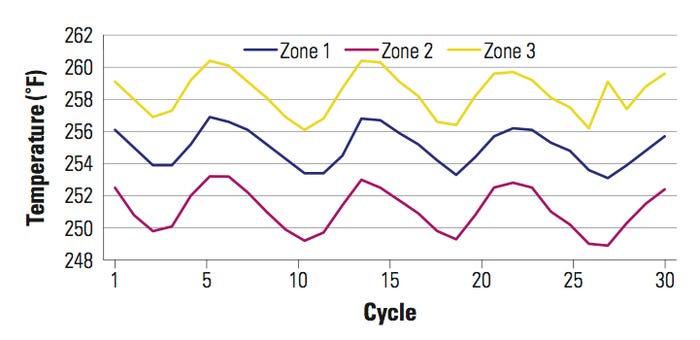
Even though it appears to have a constant temperature, the reality is that the surface temperature in the platen varies more than is being displayed. However, the fluctuations are not detectable because the RTDs are monitoring the heating block temperature and not that of the sealing surface. To illustrate this situation, consider a tray sealer with a 16 × 16-in. platen size. The platen was divided in three zones (1, 2, and 3). A temperature mapping was performed and the zones were compared.
|
Figure 2. [Click to enlarge] Temperature mapping of a 16 × 16-in. platen divided into three zones (1, 2, and 3). |
In Figure 2, the temperature set point is 265°F without any temperature offset on the temperature controller or PLC. When compared with zone 1, there is a temperature differential of 10°F between the temperature controller display and the actual sealing surface temperature. In this example, the temperature displayed on the temperature controller changed ±2°F. In reality, the sealing surface area changed by more than 2°, as illustrated in the graph. The sealing surface temperature was constantly changing in a sinusoidal manner, exhibiting up to a 4°F temperature drop on the sealing surface on each zone. In addition, there was close to a 7°F temperature difference across the platen between the zones.
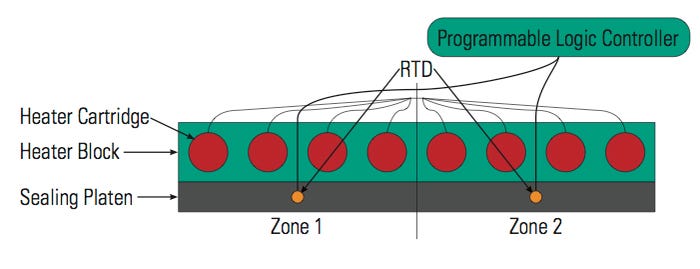
By simply using an additional RTD and changing the location of the RTDs (see Figure 3), the process can be monitored and controlled without changing the thermodynamic design.2 Having two RTDs mounted in the sealing platen enables better temperature control for the following reasons:
The temperature displayed is closer to the temperature at which the package is sealed.
The system has a better response to temperature changes across the sealing platen surface.
The heat transfer is less dependent on the environmental conditions where the sealer is located.
It is easy to detect a surface temperature problem in the event that the platen becomes loose.
It is easy to detect a problem in the temperature distribution across the platen. The PLC can be programmed to stop in the event that the temperature variation from RTD1 to RTD2 exceeds a set number of degrees.
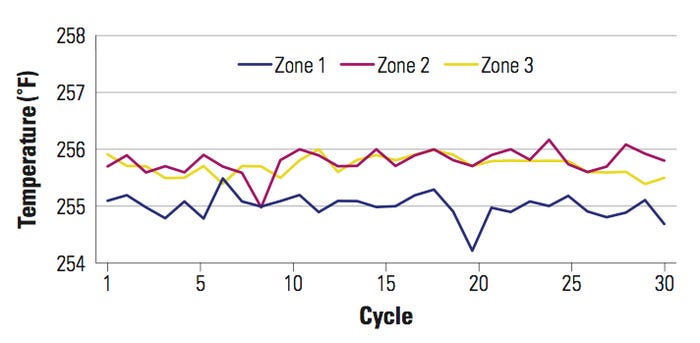
In Figure 4, the temperature set point is 256°F with a –5°F temperature offset on the temperature controller or PLC. There is no temperature differential between the temperature controller display and the actual sealing surface. This setup was achieved by having a temperature offset in the temperature controller or PLC to teach the system to display the actual surface temperature on the platen. By moving the RTDs to the sealing platen, the temperature differential between the temperature controller and the actual sealing surface temperature was reduced by half (5°F). This variation was then corrected by adding a temperature offset to the system.
|
Figure 3. [Click to enlarge] An alternative setup for controlling temperature in the platen. |
In this example, the temperature variation displayed on the temperature controller did change over time. It was ±1°F from the set point, and the sealing surface area also demonstrated the same kind of variation, as illustrated in Figure 4. The sealing surface temperature did not change in a sinusoidal way as it did in the previous example.
Dwell Time
Every machine requires a point of reference to trigger the dwell timer. Some machines have a mechanical switch while others are triggered by an output signal or sensors. Let’s evaluate each one of them.
Mechanical Switch. A mechanical switch on the side of the machine can detect the presence of the blister or tray when the tool is underneath the sealing platen. Once the switch is activated, the dwell timer starts counting. The PLC then delivers the signal to the solenoid valve. The cylinder moves down and it is kept down until the timer stops. The signal goes back to the controller and the cylinder retracts, thereby completing the sealing process.
The problem with this design is that the cylinder movement is part of the total dwell time process. If this movement starts to change, meaning that it takes less or more time to reach the package, then the process may change too. This problem can get worse if the sealer is controlling the cylinder inlet pressure with a flow control valve to prevent a pressure drop in the air compress line. The bottom line is that the process relies on an element that cannot be monitored and controlled, and therefore it is impossible to determine how much of this movement is part of the total sealing time.
Output Signal. Another option is to trigger the dwell timer at the same time that the PLC sends the signal to the solenoid valve that controls the cylinder movement. Then the cylinder moves down and it is kept down until the timer stops. The signal goes back to the controller and the cylinder retracts, completing the sealing process. In this type of setup, the mechanical switch process is eliminated but the cylinder movement is still part of the total dwell time.
Sensors. Sensors can trigger the dwell timer when the cylinder has reached a certain position. This setup can be achieved by having magnetic sensors verify the position on the cylinder. Once the cylinder moves and reaches a certain point, the dwell timer starts. This location can be established by having a standard measured pin gauge between the platen and the sealing gasket. Then, set the cylinder downward and position the sensor to activate when the platen touches the pin.
Using this system, the dwell timer will always start at the same point every time the sealer is cycled. It is not dependent on any movement or PLC signal to activate. More importantly, the dwell time reflects the actual sealing time.
Pressure
As part of every sealing process development, a surface response design of experiment (DOE) is performed. For this analysis, a low and high process parameter window is determined for each of the elements involved in the sealing process: temperature, dwell time, and pressure. Once the runs are made and evaluated based on response variables, the DOE is optimized. As part of this optimization, the sealing pressure tends to go the high end of the DOE process parameter window. Be careful about setting the process to a high-pressure condition because sealing gasket wear is not captured as part of the DOE process. High-pressure-condition processes work for a short period of time—until the sealing gasket wears out and the process outcome changes. These are considered high-maintenance processes because they require frequent gasket changes to keep the process the same. Therefore, it is important to know which pressure paramater window the DOE is set up for. One way of determining whether too much pressure has been applied to the tray flange is by visually inspecting the gasket compression during a cycle. There are other ways to calculate the force being applied by the cylinder, e.g., using the inlet pressure and the bore size, but the visual inspection is particularly helpful if the cylinder surface area is unknown.
|
Figure 4. [Click to enlarge] Temperature mapping of a 16 × 16-in. platen with the RTDs moved onto the sealing platen. |
It is also recommended that a force gauge be mounted on the sealer as part of the installation qualification process to understand the force of the cylinder based on the inlet pressure. This configuration will determine whether the proper airflow is going to the cylinder or whether there is an air leak that is preventing the cylinder from having a constant force throughout the entire sealing process.
Another variable in the system is whether the sealing process exhibits a significant pressure drop on the gauge when the cylinder is activated. Questions that should be asked are: How long would it take to overcome the pressure drop and deliver the force required based on the set point? Does the pressure drop ever recover to the set point? This problem can be caused by not having the proper airflow for the type of cylinder being used, which can lead to increased demand on the system for cylinder travel. The farther the travel, the more airflow needs to go to the cylinder. Dropping the main compress air line that feeds the sealer shared with other machines can exacerbate the situation. By adding an accumulator air tank and having a dedicated inlet pressure line to the sealer, the cylinder can have better repeatable performance from cycle to cycle.
Conclusion
As part of the equipment design, make sure that requirements are met but also note how they are achieved. Different methods of establishing, monitoring, and controlling elements such as temperature, dwell time, and pressure can affect the process outcome over time. Setting the temperature and dwell time at a certain value does not mean that the package is sealed at that particular set point. Variation may be unnecessarily added to the process depending on where the temperature is monitored and when the dwell timer is triggered in the process. It may appear that process inputs are controlled and monitored. But in some cases, they can change the sealing process, and no one knows it until a failure occurs.
There are other variables to consider such as heater style and power, enclosed versus open sealing equipment, sealing platen coatings, etc. The key to every sealing process is having the critical parameters monitored and controlled to reduce the amount of variation that can be introduced into the process. Following the machine design recommendations presented here can help manufacturers to know, characterize, and optimize their sealing processes as well as reduce variation.
References
1. ISO 11607, “Packaging for Terminally Sterilized Medical Devices—Part 2: Validation Requirements for Forming, Sealing, and Assembly Processes” (Geneva: International Organization for Standardization, 2006).
2. M Sherman, Medical Device Packaging Handbook (New York, CRC Press, 1998).
Charlie Rivera is a manufacturing engineering manager based in Largo, FL.
About the Author(s)
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
