4 Common Failure Points of Sterile Barrier Packaging Validations
A great place to start investigating a packaging test failure is with the four most common failure points encountered in sterile barrier packaging validations.
September 12, 2014
By Wendy Mach and Jennifer Gygi
|
The dark areas indicate where the seal is starting to clarify. |
Before package testing can begin, there are several items that need to be considered, including—but not limited to—cost, sample size, test method, and acceptance criteria. Arriving at the perfect combination of variables in a packaging test validation procedure is just the beginning.
But what if your packaging fails to perform as expected? The answer is simple: investigate. A test failure does not automatically mean that the packaging is inadequate. It does, however, demand evaluation and adjustments. A great place to start investigating a packaging failure is with the four most common failure points encountered in sterile barrier packaging validations.
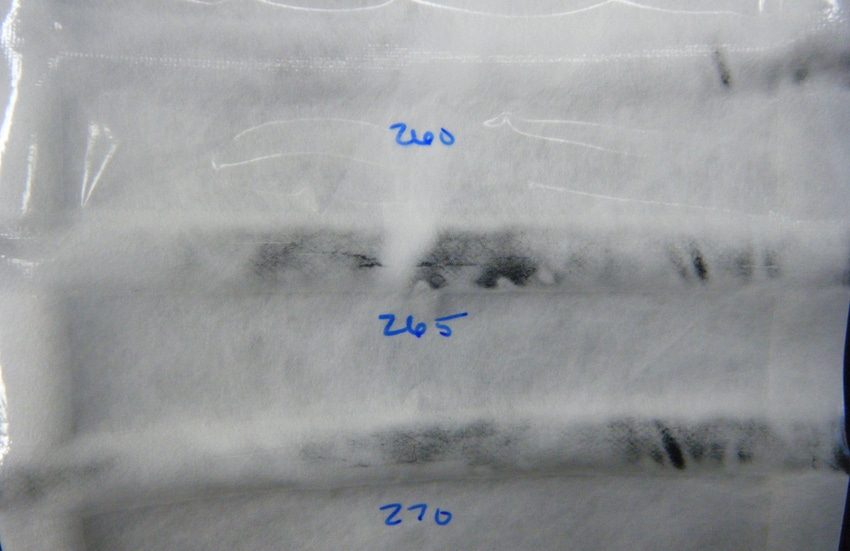
1. Oversealing. More does not necessarily mean better when it comes to sealing packages. Applying too much heat will melt Tyvek and fuse it to the film layer, causing a clarification of the materials. As sealing temperatures increase, the amount of overseal and clarification damage also increases, becoming more visible, in turn.
The image at the top of the article shows bar seals executed at three different temperatures and the associated oversealing and clarification. Oversealing can be visually identified by the presence of curling or peeling seals and spotty patches where the damage is most significant. These problems are not only unappealing; they can also cause problems with physical testing.
The amount of overseal becomes more visible as the temperature increases. Although the seal may appear to be intact, clarification of the materials can result in failures along the edges of the seal by creating tiny holes. In a seal peel test, oversealing may cause delamination of the Tyvek when peeled, which can lead to inconsistent peel values. Ensuring seals are uniform in dimension and homogenous across the width of the seal will help alleviate this type of package failure.
2. Tears/pinholes. These types of failures usually occur on the face of a package rather than on the seals. Tears and pinholes are frequently a result of the product’s natural movement in relation to the package during shipping and distribution. Packaging should be designed to keep all parts of the device from moving and shifting within the package. To avoid this problem, sharp edges should be protected with additional packaging, such as inner pouches, paperboard inserts, foam, or trays.
Additionally, the weight and shape of a device, if not accounted for in the packaging design, can cause tears and pinholes when devices are stacked in larger boxes. The compression of multiple packages stacked into a single box will cause additional stresses on a package and need to be considered. A final consideration should be given to product placement within the shipping container to avoid inadvertent damage by box cutters.
|
An example of inner package wearing on outer package after distribution testing. |
3)Packaging configuration. An initial investment in finding suitable packaging from the onset is fundamental to the success of a validation. During the product’s design phase, it is important to confirm that the product and packaging are compatible. The biggest failure point here is purchasing a pouch that is too large for the device and then folding it to make it fit into the dispenser box.
This approach is strongly advised against because folding Tyvek can cause layer separation. This allows the air coming through the porous material to pool in the separated area and emit through the channel created between the two separated layers, thus leading to bubble emission failures. If these failures are observed, ensure that the package failure is verified by performing a dye migration test. This test will either confirm a true channel failure or prove the separation of the porous substrate. The best course of action here, though, is to ensure that the sterile barrier system (SBS) is correctly sized for the device.
4. Compatibility with sterilization. You may be tempted to use a one-size-fits-all approach to packaging for your portfolio of products. But you need to consider the expected sterilization method for the product as part of the packaging design. Just as not all products are suitable for all sterilization methods, not all packaging materials are suited for all sterilization methods. It is important to verify that all materials in the package—both the SBS and protective packaging—are compatible with the chosen sterilization method. Using an incompatible material will lead to failures in the packaging validation.
Bonus tip: Thermal glass transition values (Tg). Another common mistake when performing a packaging validation is to attempt to save time by selecting accelerated aging process temperature values without considering the material’s thermal glass transition value (Tg). The glass transition of a material marks the temperature at which materials flow to a liquid state. By using overly elevated aging process temperatures, packaging materials can melt and become useless for the remaining validation testing.
Current temperature recommendations for accelerated aging of terminally sterilized medical devices for establishing the expiry date are that they should be no higher than 60°C, with 55°C as the target value. When working with temperature-sensitive materials, it is best to remain 10° to 15°C below their Tg value to prevent product from interfering with the packaging validation.
Packaging validation is no easy feat. But spending time upfront during the design phase to ensure you have selected the correct size package, chosen a shipping container that is compatible with your sterilization method, and employed the correct sealing parameters can prevent problems down the line.
Learn more about common medical packaging failures and how to deal with them in the Medical Packaging Community. |
Wendy Mach is the packaging section leader at Nelson Laboratories.
Jennifer Gygi is the packaging study director III at Nelson Laboratories.
You May Also Like


.png?width=300&auto=webp&quality=80&disable=upscale)
