MRP Reduces Costs and Shortens Lead Times for Ablation Catheter Manufacturer
April 16, 2007
Originally Published MPMN April 2007
PROFILE
MRP Reduces Costs and Shortens Lead Times for Ablation Catheter Manufacturer
M. Callan Fischer.
|
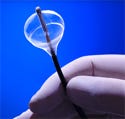
ProRhythm’s AF ablation catheter focuses ultrasound energy by use of a proprietary dual-balloon design. European rollout and clinical trials in the United States prompted the need for an MRP system. |
A medical device manufacturer in Ronkonkoma, NY, has developed a catheter-based system using high-intensity focused ultrasound (HIFU) to treat atrial fibrillation (AF). ProRhythm’s (www.prorhythm.com) AF ablation catheter focuses therapeutic ultrasound energy by use of a proprietary dual-balloon design, and represents a breakthrough treatment for millions suffering from paroxysmal atrial fibrillation.
The product can mean the difference between life and death. However, just a year and a half ago, rollout of ProRhythm’s 10-part product line in Europe led to serious obstacles.
The company needed a centralized business-management system with strong Manufacturing Resource Planning (MRP) functionality to move from R&D into full-bore manufacturing supplying the European market. Even more crucial were its ongoing FDA trials here in the United States. FDA approval of its catheter-based system represented the culmination of nine years of research and development and would allow the firm to sell to a potentially huge market—but inventory problems loomed.
“The need for an MRP system geared to medical device manufacturing was clear. We were facing rollout in Europe, plus our feasibility trials here in the United States,” says David Cichy, controller at ProRhythm. “Shortages could bring trials to a halt. Yet we didn’t want cash flow problems that come with excess or obsolete inventory. That balancing act required the help of an MRP system.”
In early 2005, Cichy spearheaded selection and implementation of ProRhythm’s new MRP II business management system, Alliance/MFG from Exact Software (Minneapolis; www.exactamerica.com).
“As a start-up prior to selling in the EU, we had never capitalized any of our inventory,” admits Cichy. “If we needed something, we bought it. That was no longer viable. We needed an MRP system to roll out the product and maintain quality. With U.S. trials already under way, we looked at a number of options, many out of the price range of a start-up. We compared Alliance/MFG with some of the other players out there and functionality jumped out: serial and lot number tracking, ease of use, unlimited-level bill of material (BOM), Crystal Reports, and integration with QuickBooks. We went with it.”
Alliance/MFG is business management software designed for medical device manufacturers that must build parts to exacting tolerances, adhere to complex customer and FDA regulations, and provide comprehensive audit trails and tracking. The software provides complete MRP-II functionality for the planning, scheduling, and business management of this type of operation. Based on Microsoft standards and using PCs running MS Windows, it offers scalability with database options such as Access, MSDE, and MS SQL.
Once implemented, ProRhythm’s new software package resulted in an efficient European rollout and smooth-running trials in the United States. Cichy likes the presence of relevant data and indispensable reports. “In the past, person A had his spreadsheet, person B had her spreadsheet. There was no centralized data. As an R&D organization, that was fine, but we’re a manufacturing firm now.”
ProRhythm’s primary device is its catheter. The company also manufactures the system or server that runs the catheter, which requires expensive metals and components, making balanced inventory critical. According to Cichy, Alliance/MFG now keeps inventory at near just-in-time levels yet averts shortages.
Also, with automated inventory and scheduling under way in 2005, ProRhythm began placing large blanket orders with key vendors, but reserved the right to move back or push forward delivery of those materials. “Going with Alliance and using blanket orders has dropped our component costs at least 10%,” says Cichy. “We input demand a year out with selected vendors. It benefits both of us—giving our vendors a long-term forecast and earning us discounts and flexibility.”
According to Cichy, the software centralizes business processes and establishes a single system of record for complying with customer and FDA regulations.
If a hospital should receive components with problems, ProRhythm must track them back to the source using lot and serial numbers. The company also tracks every single procedure involving the use of its products. They maintain records on how the procedure went, and, if after a few ablations a catheter is no longer functioning properly, they investigate.
“There is no room for error in the medical device business,” says Cichy. “We test every single lot for reliability to make sure the product meets certain specs. If it doesn’t, we scrap it. Alliance has been indispensable in this regard.”
Alliance/MFG quality assurance helps the firm conform to the tight tolerances required in the industry. Shop floor employees can identify nonconforming parts or materials and move them out of production into inspection queues for appropriate action.
Cichy relies on the system’s unlimited-level bills of material to separate out costs and pinpoint opportunities to save. One of ProRhythm’s most complicated bills of material entails 30 different subassemblies. Using customized reports plus the indented, multilevel BOM, ProRhythm has begun tracking how long it takes to make each of its products.
“We track all our labor hours and have actually been able to improve our lead times by 20%, just by looking at all the different steps and questioning why this step or that step is taking so long. For example, we might move around operators, or change the way we’re putting the product together. The result is greater speed.”
The software includes Crystal Reports, a tool that Cichy uses to customize reports. “With Crystal Reports the average person can take care of it,” he says. “I make reports into exactly what the individual wants. Sometimes we run the same information but provide it in three different formats to meet three people’s requests.”
ProRhythm now has production operation codes within every operation and generates reports that identify how long each operation takes. “This monitoring makes us more efficient,” says Cichy, who looks at various reports once or twice a month for trend data, to analyze scrap rates, cycle times, operations, and deliveries.
As for implementation, ProRhythm had little difficulty. “If you know Access, then Alliance is pretty user-friendly,” says Cichy. “You put everything in Access files and simply upload them into the system. We had one consultant do a couple of days of training, but for the most part, we implemented it 95% on our own. Nor have we ever had to call technical support. With the Access database, you simply go in, find the problem, and correct it.”
As controller, Cichy appreciates Alliance/MFG’s integration with QuickBooks. Without it, there would be a complicated reconciliation at month end. He says, “Now, when a purchase order is received, the system automatically generates a voucher for use in QuickBooks. We’re a small company with a specialized product and probably 500 vendors total, so for us that’s a huge time-saver.”
Just two years ago, ProRhythm lacked the production capacity, inventory control, tracking, and reporting to fuel its European rollout and to ensure smooth-running U.S. trials. Now with MRP in place, the company is meeting all ship dates, maintaining high quality, controlling costs, and preparing for the big push to come in the United States. “With luck, our clinical trials in the United States will be completed successfully in the next year or two,” says Cichy.
Copyright ©2006 Medical Product Manufacturing News
You May Also Like
.png?width=300&auto=webp&quality=80&disable=upscale)
