This Advice Will Help You Improve Your Device History Records
July 2, 2015
How easily can you trace every lot, batch, or unit of product? The answer is important. In fact, it will be a factor that defines your medical device company's ability to prosper.
 Julie Fraser, IYNO Advisors
Julie Fraser, IYNO Advisors
Between the FDA's requirement for unique device identifier (UDI) and the regulatory scrutiny of device history records (DHRs), traceability is always a hot topic in the medical device industry. But the challenge is that no single department or system has the entire answer. Traceability requires information from across all aspects of the operation.
Fortunately, modern information systems can reach across the organization to collect, collate and assemble the DHR automatically, without paper-- creating the electronic eDHR. But this is not a straightforward IT issue, and is one that standard enterprise IT applications such as enterprise resource planning (ERP), business intelligence (BI) and quality management systems (QMS) are not likely to be able to handle. These applications aren't designed to handle the huge volume and numerous variants of DHRs across an organization, and are typically structured around documents, which doesn't deliver any benefits beyond quality control and compliance.
The best way to generate eDHRs is by means of manufacturing execution or manufacturing operations management (MES/MOM). Much of the core DHR information comes from the production plant (e.g. dates and quantity manufactured and released, acceptance records, labels). You need systems that focus on data, versus merely engaging in document management. MES/MOM systems are first and foremost designed to provide visibility, enforcement and control to the production process. They generate these compliance records as a by-product.
Granted, having full control inside the production floor does not necessarily guarantee complete traceability will be available. So the best modern MES/MOM systems have a broad footprint to include quality and maintenance. They also can pull in data from other systems. In fact, all of the elements required in an eDHR can be recorded by this type of MES/MOM system, and that will delight auditors. Beyond that, the MES/MOM will enforce standard operating procedures to improve compliance, while also making data about the product and process available instantly.
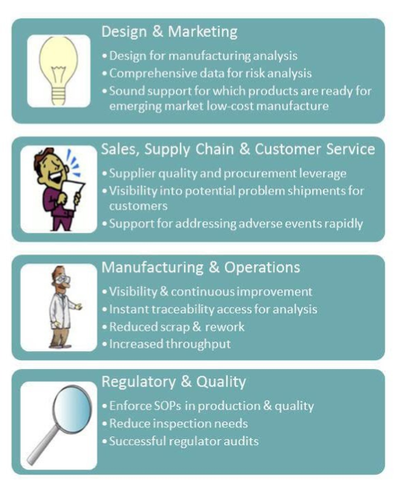
What other uses might you find for this type of system and the data it produces? Here are a few:
For planning to update actuals, materials usage and scrap rates, enable personnel scheduling and planning, financial planning and forecasting for both costs and revenues timing, and customer order promising based on a far more accurate view of what is likely to be available to ship when.
As production proceeds to guide operators, technicians, quality and engineering activities and prevent production and documentation errors; also to see production status, materials or parts problems, actual-to-plan as it changes, and performance per plant, operator, product, or piece of equipment.
After the fact to analyze for best practices, and in other departments to make critical decisions about new product design, introduction into various regional markets, outsourcing readiness, supplier and parts performance, and all sorts of weekly, monthly and quarterly reporting by operations and finance.
In predictive mode during technology transfer, joint venture planning, capital planning, new plant expansions, financial warranty reserve planning, and new product development.
So traceability can be seamless across many disciplines in the enterprise, and it can deliver them all benefits. Taken in aggregate, this becomes a company-wide effectiveness and profitability opportunity. Will you seize it?
Refresh your medical device industry knowledge at MEDevice San Diego, September 1-2, 2015. |
Julie Fraser is principal and founder at IYNO Advisors (Cummaquid, MA). Find out more from her about traceability requirements, and the role of integrated manufacturing software in eDHR and UDI compliance, in the Dassault Systèmes webinar: "Making Traceability Work for the Business: Effective eDHR and UDI," hosted by Qmed.com. For more information on how Dassault Systèmes is helping manufacturing executives within medical device companies to improve quality across their global operations, please visit Dassault's Made to Cure for Medical Device information center.
About the Author(s)
You May Also Like


