Humidity and Microbial Penetration in Porous Materials
Sterile-grade medical papers, such as Ovantex display barrier properties that are used in medical applications.
The sterility of most medical device products is of paramount importance. Sterile products are packaged, stored, and distributed in various locations that expose the packaging to a range of relative humidity levels throughout the distribution cycle. With porous packaging materials used often by the medical device industry for packaging sterile devices, maintaining the microbial barrier becomes of grave importance. Although there has been some research examining the effects of factors, such as material structure and dispersion concentration, on the microbial barrier properties of these porous materials, the impact of relative humidity is still unknown.1,2 This article attempts to correlate relative humidity levels with the amount of penetration through four porous packaging material compositions: coated and uncoated Tyvek 1073B, coated 55-lb latex-impregnated medical-grade paper, and dot-coated Ovantex.
Materials and Methods
Equipment. Achieving the desired relative humidity (RH) levels—15%, 50%, and 90%—required construction of three temporary atmospheric chambers. These chambers were built using three 20-gal aquariums and various salt solutions. Silicon grease and fiberglass were used to form an airtight seal around the top of the three aquariums. With the temperature remaining constant at 23 ± 1°C (73.4 ± 2°F), it was determined that lithium chloride, magnesium nitrate, and potassium nitrate would be the saturated salt solutions required to achieve levels close to the low (15%), nominal (50%), and high (90%) RH levels desired.3 Sper Scientific and Oakton hygrometers were placed in the environmental chambers and used to read the real-time RH and temperature values. Tie racks, purchased from Home Depot, were used to keep the samples separated and vertical during preconditioning, which would ensure that all surfaces of the sample were exposed to the environment’s moisture. Testing primarily followed the ASTM F2638 standard test method for using aerosol filtration measurement of the performance of porous packaging materials, which outlines specific configuration of equipment for the dual particle counting system used in this research.4 The equipment was configured using a nebulizer, two particle counters, a data logging system, a manometer, a pressure regulator, and a customized sample-holding fixture. All flow readings were in direct engineering units, standard liters per minute (slpm).
Material Selection and Preparation. The materials tested include uncoated Tyvek 1073B, coated Tyvek 1073B, dot-coated Ovantex, and coated 55-lb latex-impregnated medical-grade paper. The coatings on both the Tyvek 1073B and the medical-grade papers were CR27 from Perfecseal. Ovantex is coated with a dot coating exclusive to Oliver-Tolas Healthcare Packaging. Materials were provided in 12-in. square samples. Forty-five samples were cut, randomly, in 140-mm squares. Each sample was then weighed and measured for thickness in an effort to characterize each material. As outlined by ASTM F2638-07, the particulate used in testing was made of polystyrene latex (PSL) particles measuring at 1 µm in diameter.4 The product used was product 5100A Duke Scientific.
Moisture-Level Selection and Reaction Measurement. All material samples were weighed before and after environmental conditioning in an effort to understand each material’s reaction to the various moisture levels. Levels were determined using ASTM D6-01: Standard Practice for Conditioning Containers, Packages, or Packaging Components for Testing as guidance.5 Nominal relative humidity level was set from the standard condition of 50 ± 2%. The extreme conditions of low and high were set at 15 ± 2% and 90 ± 5% as desert and tropical conditions, respectively.
Measurement of Penetration. Because previous research indicates that physical (particulate) penetration performance can be correlated to that of microbiological barrier performance, research was conducted with slight adjustments to Method B in the ASTM F2638-07 test standard as a surrogate microbial barrier.6,7 Challenge dispersion concentration was set at 10,000 particles per dm-3. Variations to the method included preconditioning material samples and challenging a separate sample at each flow rate change. The decision to change samples for each flow rate was made to reduce the likelihood of drying out the preconditioned material sample and to reduce the potential for particle blockage due to a particle packed sample.
Flow rates were selected by subjecting unconditioned material samples to Method B of ASTM F2638-07 test standard.
Penetration rates were logged for samples beginning with a flow rate of 0.03 slpm. Flow rates were then effectively doubled until a peak in particle penetration was observed, followed by a continuous decrease. Flow rates were then chosen to include the approximate peak rate as well as two flow rates on either side of the peak. Tyvek 1073B coated and uncoated were tested at 0.06, 0.12, 0.24, 0.48, and 0.96 lpm. Ovantex and medical-grade paper were tested at 0.03, 0.06, 0.12, 0.24, and 0.48 lpm.
Material samples were exposed in the environmental chambers for a period of at least 48 hours. Samples were then removed from the chamber and weighed using an analytical balance. Weights were recorded and the samples were secured into the sample holder for the duration of the test (3 minutes). Two minutes were used to stabilize airflow and particulate levels, and one minute for the particulate challenge. The system recorded particle challenge and filtrate counts, flow rate, and pressure differentials every 6 seconds. Time range for the testing was recorded, so data could be extracted for analysis.
Samples were removed from the sample holder and posttesting weights were recorded. The process was replicated for all 180 samples tested following the randomized, complete block experiment design. The experiment design included three replications for each sample condition. Sample conditions consisted of various relative humidity and flow rate combinations.
Results
Moisture Reaction. The initial data collected to characterize the material samples show that all four materials remain relatively consistent in weight. However, as expected, due to the structural variations, the coated and uncoated Tyvek 1073B are highly variable with regard to thickness. Although initial weights were measured at an undetermined relative humidity (RH) level, postconditioning weights can be used to generally understand the material’s water retention capabilities. Because Tyvek is manufactured from 100% HDPE, it was not expected to respond significantly to changes in relative humidity. Both coated and uncoated Tyvek 1073B showed no consistent response to RH levels. As expected, the materials containing cellulose fibers and consistent thickness measurements, Ovantex and medical-grade paper, demonstrated fairly consistent responses at each of the tested relative humidity levels. The cellulose fibers are more likely than HDPE to absorb or desorb the moisture in the RH chambers, which allows the materials to show more consistent responses to change in RH levels.
|
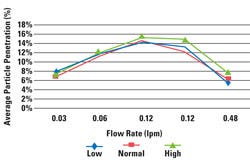
Figure 1. The average percent penetration of coated 55-lb medical-grade paper at low-, nominal-, and high-humidity conditions. |
Microbial Penetration Test Results. The equipment used for the testing recorded particle penetration every 6 seconds through the duration of the experiment, which resulted in 10 readings per sample. Each of the 10 readings was averaged to produce the particle penetration percentage for that specific material and sample condition. There were three replicates for each sample condition. The penetrations were averaged to produce the average percent particle penetration for the given material at a specific flow rate and relative humidity level.
The average percent penetration through each material was plotted and analyzed. As an example, Figure 1 shows the resultant curve for coated 55-lb paper. Initial curves for some of the materials showed distinct microbial penetration versus RH level curves with values varying as RH levels changed. These observations led to the expectation that RH levels did significantly affect the microbial penetration for both the coated Ovantex and the coated medical-grade paper materials. Both coated and uncoated Tyvek 1073B demonstrated much variability in the microbial penetration data at all RH levels, which led to a hypothesis that neither the coated nor uncoated Tyvek 1073B would result in a microbial penetration rate that was significantly dependent on RH levels. Results for both Tyvek types did demonstrate a statistical difference at the penetration percentages observed, but did not show a practical significant difference.
Using statistical analysis software (SAS) to perform a general linear model (GLM), these hypotheses were tested for statistical significance. The statistical significance level selected was a p-value of 0.05. This means that, using a GLM, if the p-value is less than or equal to 0.05, then 95% of the time a correlation exists between the two variables being tested. The GLM analysis concluded (p-value greater than 0.05) that the flow rate through the sample and the RH level affect the material’s microbial penetration rates independently of one another. This was the case for all four materials. This means that the two dynamic variables of the test—flow rate and RH level—are independent of one another. Because they are independent, further analysis was done to test both variables for their relationship to the microbial penetration properties.
Using the same GLM analysis techniques, a study was performed with respect to the two dynamic test variables and microbial penetration properties. The conclusion of these tests was that the flow rate across the sample was only found to be significant (p = <0.0001) for the medical-grade paper and Ovantex. RH was found to have a significant effect (p = 0.0016) on the microbial penetration properties for medical-grade paper (see Table I, p. 38).
Discussion
Flow rate results found in the medical-grade paper and the Ovantex were expected, as earlier research has observed similar occurrences.6 This earlier research indicates a significance (p = 0.05) of flow rate on microbial penetration for several materials. The current research shows a significance (p = 0.10) between the flow rate and particle penetration for the uncoated Tyvek 1073B. Further research would be necessary to determine whether it was the differences in test method or the known structural variations in Tyvek that caused the slight change in confidence for the flow rate and microbial barrier relationship. It is recommended that future research characterize the structural densities and porosities for individual Tyvek samples. This characterization will make it possible to determine whether observed inconsistencies or statistically insignificant findings are due to actual test parameters or variations in the materials’ structure.
Because little research has been published on the effects of coating on Tyvek, the findings of this research are interesting. This research suggests that the addition of Perfecseal’s CR27 coating to Tyvek 1073B significantly decreases the impact flow rate has with respect to microbial penetration. Future research examining various coatings with respect to their effect on microbial penetration and their effect on properties strongly related to microbial penetration (i.e., flow rate) could prove to be beneficial.
Because the cellulose fibers that form paper are susceptible to moisture, it was not surprising to find that relative humidity had a significant effect with regards to the microbial penetration through medical-grade paper. It was interesting that the Ovantex, which also contains cellulose fibers, showed no significant relationship between microbial penetration and RH levels. Because the exact makeup of Ovantex is unknown, it is impractical to make suggestions about the effect that cellulose fibers and RH have on microbial barrier properties, however much research can be performed in this area. It would be interesting to determine what types of fibers are most susceptible to changes in RH levels, and what percentage of cellulose fibers in a material structure compromise its microbial barrier properties. It would also be interesting to research how materials containing cellulose fibers react to microbes over time. Could microbes with a food source (cellulose fibers and water) eventually penetrate a package by eating through the material?
Conclusion
RH levels have a significant effect on the particle penetration levels for the medical-grade paper tested, 55-lb latex-impregnated paper coated with Perfecseal’s CR27 coating. Manufacturers planning to package using medical-grade paper should be concerned with the distribution and storage relative humidity levels for their products, because ISO 11607 requires manufacturers to ensure the sterility of their product from production until point of use.
RH levels do not appear to have any effect on the particle penetration levels for the dot-coated Ovantex, by Oliver Medical, or DuPont’s coated and uncoated 1073B Tyvek.
References
1. A Tallentire and CS Sinclair, “Variations in Structure and Microbial Penetrability of Uncoated Spunbonded Polyolefin (Tyvek),” Medical Device + Diagnostic Industry 6, no. 7 (1984): 57–61.
2. A Tallentire and CS Sinclair, “The Influence of Dispersion Concentration on Microbiological Barrier Performance of Porous Packaging Materials.” Medical Device + Diagnostic Industry 8, no. 8 (1986): 34–37.
3. L Greenspan, “Humidity Fixed Points of Binary Saturated Aqueous Solutions,” Journal of Research of the National Bureau of Standards—Physics and Chemistry 81A, no. 1, January–February (1977).
4. F2638-07, “Standard Test Method for Using Aerosol Filtration for Measuring the Performance of Porous Packaging Materials as a Surrogate Microbial Barrier” (West Conshohocken, PA: ASTM International, 2007).
5. F4332-01, “Standard Test Method for Conditioning Containers, Packages, or Packaging Components for Testing” (West Conshohocken, PA: ASTM International, 2007).
6. A Tallentire and CS Sinclair, “A Discriminating Method for Measuring the Microbial Barrier Performance of Medical Packaging Papers,” Medical Device + Diagnostic Industry 18, no. 5 (1996): 228–241.
7. A Tallentire and CS Sinclair, “Definition of a Correlation between Microbiological and Physical Particulate Barrier Performances for Porous Medical Packaging Materials,” PDA Journal of Pharmaceutical Science and Technology 56, no. 1 (2002): 11–19.
Jennifer L. Blocher is medical device applications specialist at Sealed Air Corp. (City of Industry, CA); Duncan Darby is associate professor at Clemson University (Clemson, SC); Kay Cooksey is the Cryovac endowed chair at Clemson; and Patrick Gerard is a professor at Clemson. Laura Bix is an assistant professor at Michigan State University.
About the Author(s)
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
