MD+DI's Manufacturer of the Year: Johnson & Johnson
It may seem an easy choice to select the largest, most successful medical device firm as the manufacturer of the year. Johnson & Johnson is broadcloth in the medical device industry, reaching consumers, patients, and physicians on multiple levels and in multiple venues—a cardiovascular surgeon who uses a Thermocool SmartTouch contact force sensing catheter in the operating room is also likely to use a Band-Aid on her son’s grazed knee. But believe it or not, there was considerable debate about whether to choose the juggernaut firm.
October 27, 2011

|
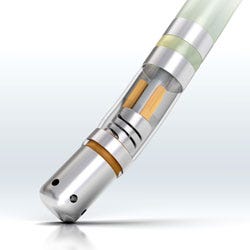
The Thermocool SmartTouch contact force sensing catheter is manufactured by J&J subsidiary Biosense Webster. It is available in the Europe. |
It may seem an easy choice to select the largest, most successful medical device firm as the manufacturer of the year. Johnson & Johnson is broadcloth in the medical device industry, reaching consumers, patients, and physicians on multiple levels and in multiple venues—a cardiovascular surgeon who uses a Thermocool SmartTouch contact force sensing catheter in the operating room is also likely to use a Band-Aid on her son’s grazed knee. But believe it or not, there was considerable debate about whether to choose the juggernaut firm. Was it too obvious? Maybe. Were we overlooking smaller firms? Probably. Was J&J’s record exemplary? Not by a long shot. But the company is still extraordinary.
But taken in context with trends in the medical device market, we realized that the only firms with spotless records are start-ups. Further, the past few years represent the first time under the current regulatory structure that macroeconomic pressures have significantly influenced the healthcare market.
As with any large company, J&J has seen its share of scrapes and successes. Luckily, the successes have clearly outweighed the missteps, or we wouldn’t be having this conversation.
So why now? What actions of this mammoth company set it apart in 2011? Well, first, this year marks the firm's 125th anniversary. The other reasons are ones that you’ve likely already guessed—J&J changed the game in 2011 in ways that no other firms were able to do. In April, it made the largest acquisition of the year with its purchase of Synthes. In one (expensive) swoop, the firm cornered the orthopedics and trauma market, staking a claim that the sector will define the company for years to come.
Then, in June, the firm shocked industry with its decision to exit the drug-eluting stent (DES) market and halt production of its once-gloried blockbuster device, the Cypher sirolimus-eluting stent. The decision signifies that J&J is willing to make tough decisions in a struggling market.
Those bold moves made for big headlines in 2011 and make J&J the clear choice for MD+DI’s manufacturer of the year. This is the inside scoop on how it got there.
New Leaders and a New Philosophy
“You expect big companies to think big, and that’s what J&J is doing,” says Richard Lincoff, one of MD+DI’s editorial advisory board members. “While other companies are becoming more conservative with resources, J&J is willing to step out.”
Lincoff gives credit for those bold moves to two new leaders at J&J, Alex Gorsky and Sheri McCoy. “The company is giving the reins to a coleadership that promises to make fearless decisions,” he says.
In January 2011, McCoy and Gorsky were appointed as vice chairs of the executive committee, reporting directly to CEO William Weldon. Gorsky continues to serve as worldwide chairman of the medical devices and diagnostics group, in addition to his role on the executive board.
With the appointments of Gorsky and McCoy, J&J has two valuable leaders ready to take charge, although Weldon has gone on record to say that he has no plans to retire just yet.
|
Alex Gorsky serves as J&J's executive committee vice chair and worldwide chairman of the medical devices and diagnostics group. |
Gorsky has been a leading voice in the industry, serving on AdvaMed’s board. His experience in the pharmaceutical industry has served to hone his skills for taking on the increasingly challenging medtech industry. Gorsky says that he and McCoy bring key perspectives to the leadership at J&J, namely an increased emphasis on finding a balance between centralized and decentralized processes, increasing innovation, and rethinking J&J�’s approach toward emerging markets.
Gorsky explained his philosophy in Ernst & Young's Pulse of the Industry, 2011. “We must accelerate the pace of experimentation with truly novel approaches to the way we do business . . . and we must quickly and broadly adopt those that prove most effective instead of hanging on to the status quo,” he said in the report.
J&J’s actions in 2011 have certainly proved that Gorsky and company are willing to put that philosophy to the test. “We’ve made some significant decisions to shape our portfolio of businesses,” he says. One of those decisions in 2011 was a big engagement.
Put a Ring on It
When proposing marriage, a groom is expected to spend about three months’ salary on the ring. For its engagement to Synthes, J&J offered an entire year’s revenue. That’s a $21.3 billion diamond. It’s not only the largest medtech acquisition of the year and the third largest acquisition in industry for the past decade, it’s J&J’s largest acquisition ever, eclipsing the firm’s purchase of Pfizer’s consumer business for $16.6 billion in 2006. On December 15, Synthes stockholders will vote on the merger agreement.
In addition to the hefty price tag, the buy is very different in tone from other acquisitions that J&J has been involved in. Normally, the company purchases smaller companies and allows the firms to operate somewhat independently. However, with Synthes, J&J immediately said it would put the business into DePuy. The plan to combine Synthes with DePuy is ambitious, and industry will look to see how well the merger is handled.
The merger helps fill a significant gap in J&J’s portfolio; it provides breadth and scale in the trauma market, particularly in emerging countries. “It is consistent with our long-term strategy to strengthen our leadership position across important healthcare markets—in this case, the $37 billion global orthopedics market,” Gorsky notes.
He says Synthes offers a huge opportunity in emerging markets. “By definition, trauma is unplanned surgery,” Gorsky says. “In emerging markets, trauma tends to mean orthopedic surgery. Many emerging countries don’t have discretionary surgical procedures. As these countries build more and drive more automobiles, for example, it will increase the need for trauma devices to help patients.”
Synthes earned $2.69 billion in sales of trauma products in 2010 and saw 10% growth in total sales in the first half of 2011. Its primary trauma products are implantable screws, nails, and plates.
It is worthwhile to note that Synthes does not produce metal-on-metal (MOM) hip implants, a product DePuy has been struggling with. Clinical evidence has shown that MOM implants exhibit high revision rates. Gorsky admits that the MOM problem is concerning to patients, families, and physicians. “We are committed to working with patients and their health insurers to address medical costs directly associated with the recall and to providing information and updates in a timely manner," he says.
An industry expert, who asked to remain anonymous, conjectures that as the acquisition moves forward, DePuy might become a smaller name, while Synthes’ products take a larger role in J&J’s orthopedics offerings.
Derrick Sung, a medical device analyst for Bernstein Research, agrees that Synthes represents an opportunity for J&J to break into emerging markets. “Synthes’ foothold in emerging markets allows J&J to develop a physician base to whom they can offer more sophisticated ortho products like hips and knees, as those economies develop,” he told Barron’s.
Gorsky acknowledges the need for J&J to gain a stronger foothold in these markets. “We’ve strengthened our global presence, especially in emerging markets like China and India,” he says. “Earlier this year, for example, we opened a new medical device and diagnostic Asia-Pacific innovation center in Suzhou, China, focused on developing products specifically for the S2 [secondary supply curve] market in China and India, comprised of hundreds of millions of people who are now gaining some degree of healthcare coverage.” In fact, Gorsky spoke to me less than a day after he got back from Shanghai.
The challenge for the merger will be integrating the sales forces of Synthes and Depuy, which have dramatically differing styles according to the Wall Street Journal. DePuy’s approach is in situ, so to speak—they are more likely to be in the physician’s office or hospital. Synthes looks to enlist physicians to teach surgical techniques. The combination of the styles could create a sales force that can’t be matched. “If they integrate the sales force correctly, it will mean unprecedented capabilities. The impact will be greater than one-plus-one-equals-two,” Lincoff says.
Gorsky notes that the company has a full integration planning team in place. “A combination of DePuy and Synthes will be well positioned to succeed in this new environment, offering surgeons and patients an unparalleled breadth and depth of technologies,” he says.
Another acquisition J&J made this year is designed to address the changing healthcare landscape. J&J subsidiary Ethicon Endo-Surgery Inc. acquired SterilMed in September for an undisclosed amount. SterilMed reprocesses and repairs medical device equipment. The buy provides vertical services to help keep hospital costs low. According to Gorsky, the move “better aligns us with our customers, who are searching for cost-effective solutions.” Gorsky says SterilMed will remain a standalone company that collaborates with Ethicon and other J&J businesses.
On the acquisition front, J&J seems to be making decisions that further diversify its already broad product lines in ways that both react to and anticipate market forces.
Shedding Skin
Most medical device companies are very good at creating new products. New lines of devices drive excitement, pull marketing attention, and reinforce branding. Good medical device companies know how to make new products. Most companies, regardless of the industry they serve, however, are less successful at shedding product lines.
But in this new economy, restructuring and taking a hard look at product offerings may be key to a company’s survival. “Restructuring is difficult under any circumstances,” Lincoff notes. “Costs aside, you have people who are committed to the market. And the stakes are doubled in healthcare because patients are relying on your care.”
So when J&J decided to eliminate its drug-eluting coronary stent, a technology that the firm pioneered, it took industry by surprise. But after the initial curveball, many experts began to realize the strength of elimination.
“J&J had good reasons to make this decision,” Lincoff says. He explains that the stent market is becoming more mature and stable. He also says there is simply not much differentiation among stent products. “They must have been asking themselves, ‘How much more can we squeeze out of this product line?’” Lincoff says.
Gorsky agrees that the dynamics of the stent market have changed and notes that demand, pricing, reimbursement, and regulatory requirements for new technologies are becoming more challenging obstacles. “Today, we believe there are other areas of the cardiovascular market where there are greater unmet medical needs and greater opportunities for growth. We remain committed to the treatment of cardiovascular disease, and we see a long, bright future in this important market,” Gorsky says.
J&J seems to be moving deeper into heart valves and ventricular assist devices. Gorsky points to J&J subsidiary Biosense Webster, which provides advanced diagnostic, therapeutic, and mapping tools used to treat irregular heart rhythms. The image on the cover of this issue is the Thermocool SmartTouch contact force-sensing catheter from Biosense Webster. This device is available in Europe, but has not yet been approved for use in the United States.
Port in a Storm and Shifting Policy
J&J’s stability and its ability to make bold moves have made it a Wall Street darling in the last few months—and with good reason. “This is the first time we’ve seen a correlation between macroeconomics and healthcare,” says Gorsky. Given that dynamic, it’s not surprising that investors and advisors would look to a calm investment port. “Our diverse array gives us good line of sight,” Gorsky notes. But it’s not just J&J’s size and diversity that have affected stock prices. Sung predicts that the Synthes acquisition and J&J’s exit from the drug-eluting stent business will take the company from 2% growth to 5–6%.
Nonetheless, the sluggish economy and shifting healthcare policy are pain points, even for J&J. “We recognize that many of the challenges brought on by the current economic downturn—lower prices and stricter utilization standards, for example—are probably here to stay,” Gorsky says.
However, Gorsky seems energized rather than undone by challenges to the industry. For example, he says that J&J supports and is committed to healthcare reform. “We thinks that increasing access for patients so they can get new innovations is absolutely essential.” That being said, Gorsky notes there is room for improvement. “There are some components of healthcare reform that need to continue to be revised,” he says, citing the device tax, which he notes could lead to unintended consequences, such as price shifting.
Regardless, Gorsky sees that healthcare policy as a cost of doing business. Policy aside, he says J&J should focus on increasing the growth of its core brands, increasing new innovations, and expanding into new markets. “This is a better use of our time,” he says.
J&J continually seeks to improve its supply chain. “Our company is decentralized, and that helps us be looser to our individual markets, but [we] recognize that certain functions, particularly in the supply chain, might be better centralized.” He notes that a consistent system helps standardize delivery approaches and share best practices.
Whatever the issue, it will be measured against the scope of services and products that J&J provides. “The breadth of our businesses gives us a view of the entire healthcare landscape and enables us to make better choices than if we were in just a handful of market segments. We see the whole field, and we make our choices accordingly.”
The real luxury is that J&J is able to invest, grow, and shed skin because of its size and financial strength. The company can simultaneously leverage slow-growth businesses and geographies to fund high-growth opportunities.
That is attractive to partners and investors, which enables the firm to “pick and choose the best opportunities,” Gorsky says.
Leaning Forward
Gorsky understands that the continued success of J&J is its ability to “lean forward,” as he puts it. In leaning forward, it may be that J&J has some shake-ups in its future. With Gorsky at the reins of medical devices and McCoy heading up pharmaceuticals, it may be that Johnson &Johnson will split its business, perhaps in a manner similar to Abbott, which recently divided its pharmaceutical and medical products businesses into two separate companies. Gorsky, however, isn’t looking for an immediate change, whatever it may be. “We are committed to our business model,” he says, pointing out that it is a model that has worked well for 125 years. Further, Gorsky notes that the diversity of the company enables better service to patients in treating disease states. “In diabetes, for example, there is no magic bullet to manage that condition,” he says. “We can take a multifaceted approach, such as bringing together biologics and devices.”
Gorsky also says that J&J’s diversity allows for career opportunities. He points to a program in which employees are invited to sit on panels within the company and share their expertise. “A business is like a garden—in order to thrive, it needs to be regularly worked, pruned, and planted,” he says.
About the Author(s)
You May Also Like


.png?width=300&auto=webp&quality=80&disable=upscale)
