U.S. IVD Industry Preps for a Comeback
Despite reimbursement and regulatory challenges, the outlook for the U.S. IVD industry seems rosy, thanks to molecular diagnostics, boomers, and Obamacare.
May 13, 2014

Although in vitro diagnostics (IVD) companies continue to grapple with regulatory and reimbursement obstacles, factors such as breakthrough technologies, a swelling aging population, a rebounding economy, and even the Affordable Care Act (ACA) are contributing to a relatively rosy outlook for the U.S. IVD sector. In fact, these positive forces are expected to boost the U.S. IVD market to more than $28 billion in 2018, according to “U.S. In Vitro Diagnostic Test Markets,” a recent report published by market research firm Kalorama Information.
Valued at $24.1 billion and accounting for roughly 44% of the global IVD market, the U.S. IVD market faces an array of exciting opportunities and daunting challenges in the years ahead. Of course, doubling as both a major opportunity and a significant challenge is the ACA. While many medical device companies are apprehensive about the potential impact of Obamacare, the U.S. IVD industry is gearing up for growth.
“IVD benefits more than most medtech industries from the ACA,” Bruce Carlson, publisher of Kalorama Information, told MD+DI earlier this year. “It is still affected by the negative portions, as IVD test instruments are considered a device and taxed. But it’s one of the few areas of medical devices that will truly see a benefit from the expected patient volume. As more people gain insurance coverage and take advantage of wellness programs and preventative care, test volumes should increase.” With that in mind, Kalorama predicts that the ACA will contribute average annual growth of 1.2% to the U.S. IVD market between 2012 and 2022.
Market Share
The top 12 companies by estimated 2013 revenue account for approximately 60% of the total U.S. IVD market, according to Kalorama. And Roche continues its reign.
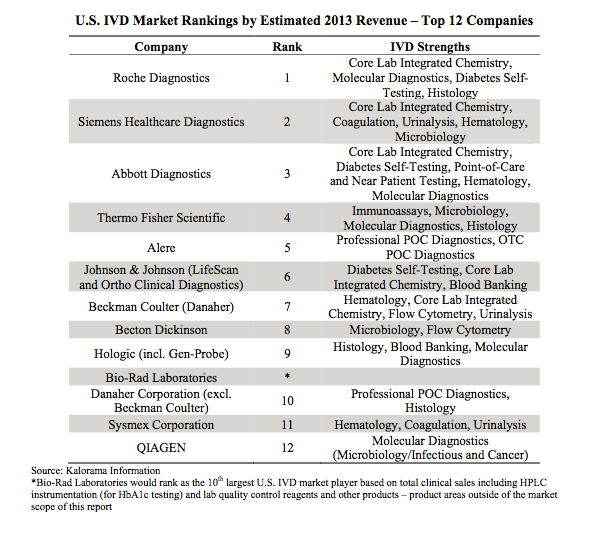
Since the release of the report, however, Johnson & Johnson made headlines when it sold off its Ortho-Clinical Diagnostics business for approximately $4 billion to The Carlyle Group. It remains to be seen how this deal could affect the U.S. IVD landscape.
The Infectious Disease Market
Market opportunities abound in the infectious disease testing space—particularly for sexually transmitted disease (STD) testing, according to Kalorama’s report. Based on such factors as the technological maturity of testing, demand, potential market size, and government support, the company identifies HIV/AIDS as the leading infectious disease testing market in the U.S.
“Other STDS,” HPV, and hepatitis also landed on Kalorama’s ranking of U.S. infectious disease opportunities in the third, fifth, and sixth slots, respectively. “In the United States, chlamydia and syphilis have two of the highest incidence rates among positively trending infectious diseases,” the report states. “Combined with gonorrhea, HPV, HCV, and HIV, STDs represent the leading infectious disease market opportunity in the United States.”
Securing the number-two spot on Kalorama’s list, however, was hospital-acquired infections (HAIs). Changes to reimbursement in 2008 in an effort to curb instances of HAIs and take costs out of the healthcare system prompted an uptick in patient screening, most commonly for methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE).
“Patient screening for MRSA is mandated by several state governments and more than 75% of healthcare facilities in the United States now conduct active surveillance testing to detect patient MRSA infections,” according to the report. “The U.S. HAI molecular testing market is expected to grow by low double digits in the coming years as MRSA surveillance is further expanded and additional HAI pathogens are included in patient screening procedures.”
Leading Market Segments
The report states that point-of-care (POC) testing represents the largest U.S. IVD market segment; POC (professional) is a $3.6-billion market while POC (over-the-counter) diabetes represents a $4.3-billion market. The immunoassay markets—both infectious and “other”—are also strong.
But today’s in-demand market segments don’t necessarily represent the growth opportunities for tomorrow. Core IVD market segments will likely experience limited—yet steady—growth. However, Kalorama states that histology, molecular diagnostics, and microbiology (molecular) are among the fastest growing market segments with a predicted CAGR of 9.3%, 8.2%, and 6.4%, respectively, to 2018. POC (professional) also demonstrates solid growth potential, according to Kalorama, at 4.5%.
“The leading growth rates of molecular diagnostics and histology in the U.S. IVD market is prefaced on their utility in cancer detection and monitoring,” the report states. “Cancer represents one of the most burdensome diseases on the U.S. healthcare system and its prevention is a leading priority.”
Learn more about market trends at the MD&M East conference and exposition, held June 9-12 in New York City. |
—Shana Leonard, group editorial director, medical content
[email protected]
[Top image courtesy of renjith krishnan/FREEDIGITALPHOTOS.NET]
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
