Antimicrobials: The Silver Lining for Infection Control
Known for its antimicrobial properties, silver is widely used in implantable device coatings to prevent bacterial growth and ward off infections. Tubing that contains a silver ion coating or lining prevents bacteria and mold from growing. It has been proven safe and effective in fighting a wide range of microbes.1
September 24, 2010
|
Luer producs containing no PVC, BPA, DEHP, or plasticizers. |
Silver is the most widely used antimicrobial. It naturally and continuously resists the growth of bacterial microbes, mold spores (fungi), yeast, mildew, algae, and biofilms. Depending on the microorganism, silver ion antimicrobial technology has been shown to initially reduce microbial populations within minutes and to maintain optimal performance for years. It does this by passing through the cell wall to block pathogenic cells from replicating.
For silver ions to be used in antimicrobial technology, they are combined with a carrier substance that is then incorporated into, or onto, virtually any material at the time of manufacturing. Materials include plastics, silicones, rubber, and all of the newest polymer blends. Silver ion incorporation into material compounds themselves is highly flexible because it allows the precise development of custom antimicrobial tube or device material blend for optimal protection. Silver provides a durable, nonleachable antimicrobial that does not result in the development of resistant bacterial strains.
Other antimicrobials are being developed that include a new class of synthetic compounds called polyalkanals with broad-spectrum activity against bacteria and viruses. They show promise for many medical tubing applications when incorporated with various new polymers. Iodine, another antimicrobial, although good upon its first application, is less effective than silver because its antimicrobial strength dissipates quickly.
Growth in Antimicrobial?Tubing
Issues such as hospital-acquired infections (HAIs), which are infections associated with urinary catheters, blood lines, heart bypass, and certain elective surgeries, are on the rise. According to the Agency for Healthcare Research and Quality (AHRQ) these HAIs cost $1.5 billion a year to treat. As of 2008, CMS and most other insurers, no longer pay for treatments of infections deemed “preventable medical errors” that occur during or after hospitalization.
The CDC recently reported that in American hospitals alone, HAIs account for an estimated 1.7 million infections and 99,000 associated deaths each year.2 Of these infections,
32% of all HAIs are urinary tract infections.
22% are surgical-site infections.
15% are pneumonia (lung infections).
14% are bloodstream infections.
Preventing HAIs requires consideration of a wide range of options, not the least of which are strict health worker training and sanitation guidelines. But because so many of these infections are tied to various the types of tubing used in a wide variety of medical and surgical treatments, adding a level of security to the tubing itself is a clear path for additional safety precaution. Tubing delivers drugs, fluids, nutrition, and oxygen to patients and removes waste products from the body. These catheters, tubes, and drains provide entry vehicles for bacteria and other pathogens regardless of the care taken by healthcare workers and the cleanliness of the facility.
|
Antimicrobial filltings and tubing with silver lining. |
Using silver-coated or -lined catheters for patients significantly reduced the most common HAI, according to a study published in the April 2008 issue of Urologic Nursing, the journal of the Society of Urological Nurses and Associates.3
Catheter-related bloodstream infections are also common within intensive care units. Many result in patient death. If this weren’t bad enough, the bugs are getting stronger. Bacteria such as methicillin-resistant Staphylococcus aureus (MRSA), also known as the “superbug,” are on the rise. Infection is also moving beyond invasive tubing use into the area of surgical implantation. This is causing device manufacturers to look for better bacteria-fighting coatings and additives to incorporate in their products.
When incorporated into medical tubing and devices, silver ions actually attack bacteria cells, choke off their oxygen supply, and keep them from multiplying. In laboratory testing, silver ion infusion has proven to be highly effective against many of the bacteria, yeast, fungi, and molds. Because silver is inorganic, it reduces the risk of antibiotic resistance.
Silver ion coatings and linings in medical tubing, connectors, and devices will kill at least 99.99% of the six common pathogens known to cause catheter-related bloodstream infections. These coatings also inhibit microbe cell reproduction.
|
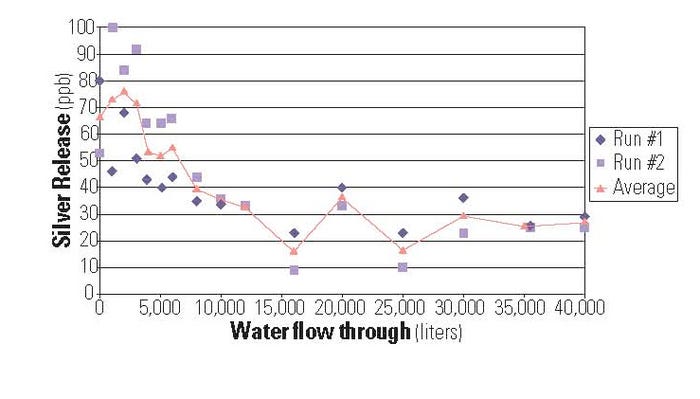
Figure 1. Level, sustainable efficacy can be found with silver antimicrobials. |
In one study conducted by an independent supplier, lifetime testing was run to generate data showing silver release and bioefficacy after tap water was run through tubing treated with silver ions (see Figure 1). Samples with a proprietary percentage of antimicrobial loaded in the inner layer with a wall thickness of 0.015 in. had 40,000 L of tap water run through them. Silver release and bioefficacy testing were carried out at regular intervals during the test.
Bioefficacy testing showed that after 40,000 L of tap water had flowed through, the tubing was still providing up to 6 log reduction in E. coli. Performance against other bacteria, and under other operating conditions (different liquids, flow rates, fluid sitting in the tubing for extended periods, exposure to harsh cleaning regimens, etc.) may vary. Based on the results obtained in this study, it can be predicted that the silver tubing tested will remain efficacious for up to 40,000 L of use under operating conditions likely to be seen in dispensing applications.
A Word About Biofilms
Although not all biofilms are harmful, those that are can wreak havoc. Biofilms are a combination of bacteria and sugars, proteins, and even DNA exuded by bacteria. Biofilms are on all surfaces with moisture. In many cases, if they establish a foothold, they can cause life-threatening infections. Biofilms can form on the surfaces of artificial hearts, stents, urinary catheters and central lines, contact lenses, intrauterine devices, joint implants, and dental implants—basically any invasive device, tubing, or implant. The problem is that biofilm formation requires strict prevention because once established, it is extremely hard to eradicate.
According to the Centers for Disease Control and Prevention (CDC), many hospital-acquired, or nosocomial, antimicrobial-resistant infections can be attributed to the formation of biofilms on implanted medical devices.2 Biofilms occur when organisms secrete a sticky substance that enables the organism to adhere to a surface. The biofilms then synthesize a polysaccharide that envelops the organism, protecting it from the body’s defense system.
Of the estimated 1.7 million HAIs in the United States annually, the CDC estimates at least 250,000 of these cases are line sepsis. With an attributable mortality rate of 12–25%, this infection is the leading cause of death associated with HAIs, second only to pneumonia. Line sepsis can lead to acute respiratory-distress syndrome, kidney failure, shock, and other potentially fatal ailments. Additional treatment costs for line sepsis average about $35,000 per infection.
Clearly, preventing sepsis must be a major priority to protect patient lives and help corral rising healthcare costs.
Silver ions provide a broad-spectrum antimicrobial action that is well tolerated by surrounding tissues. Plus, this antimicrobial action offers the benefit of being compatible with most medical device manufacturing materials. Silver ion–lined antimicrobial tubing and fittings provide added defense against biofilm formation. The antimicrobial coextrusion activity inhibits bacterial colonization.
A recently completed a study of the effect of one silver ion material on biofilm growth was aimed at medical
|
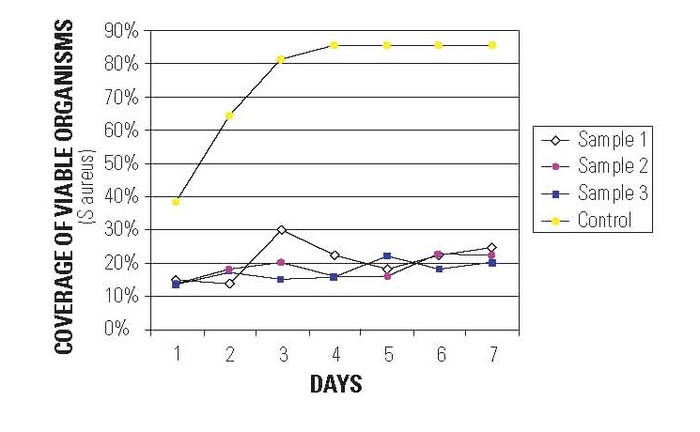
Figure 2. The benefits of antimicrobial protection. |
applications involving indwelling devices such as catheters that contact living blood and can promote infection. The study was performed at an independent laboratory. It used a specially designed biofilm reactor using Staphylococcus aureus in a sheep blood medium and the test ran for 7 days continuously. The catheters were evaluated using confocal microscopy that can observe both area coverage of the organisms on the catheter surface and whether the bacteria are alive or dead (see Figure 2). The final results of the study indicated that the control catheter became highly colonized with viable organisms within a day or two, while the treated catheters exhibited low viable organism coverage.1
Antimicrobial tubing and fitting products are manufactured in a cleanroom environment and as an added precaution, antimicrobial tubing is used on extrusion lines. As the tubing is made, it progresses through a water bath that cools the tubing before it gets to the cutter. This water must be clean so that it does not contribute any bioburden. Biofilm can easily form on the sides of the tubing that keep the water circulating through the bath. To ensure that it is kept clean and doesn’t contribute to the bioburden, antimicrobial tubing is used in conjunction with several water sterilization techniques to inhibit bacterial growth in all tubes during manufacturing.
New Materials Provide a Clear Advantage
Although it is a low-cost, viable material—and very well suited for many medical applications—PVC has decreased in popularity, and many tubing and medical device manufacturers are looking for alternative materials. This decline is due to both recycling and ecological issues and recent FDA warnings of the potential health risks of DEHP use in the production of many PVC-based products.4 Aside from these issues, PVC materials tend to darken when antimicrobial coatings are applied to them. Althought the color change does not affect the usefulness of the tubing, most healthcare workers want clear tubing, even with antimicrobial linings. Silicone materials have proven hard to work with in some applications and hold the potential for allergic reactions in patients and medical workers. Because of these and other issues, some antimicrobial tubing and device providers are now moving away from strictly offering PVCs and silicone materials and moving toward the use of newer polymers.
|
Cleanroom-manufactured fittings moving through the quality control process. |
Also, new functionalized polyolefins are being marketed by developers. A range of ethylene-acrylic ester copolymers that meet the very specific medical tubing needs, from IV fluid and blood bags to catheters and infusion lines, are now available. These materials offer intrinsic flexibility, wide-range sterilization compatibility, and easy processing. But, the list of new materials could fill a book by itself; acetal copolymers or polyoxymethylene (POM), polybutylene terephthalate (PBT), liquid crystal polymer (LCP), and polyphenylene sulfide (PPS) are only a few, and new blends are continually being developed by polymer manufacturers.
Some promising polymers in the area of antimicrobial medical tubing and fittings are thermoplastic elastomers (TPEs). TPE is a new RoHS-compliant plastic material that combines the processing advantages of a thermoplastic with the properties and performance of a rubberlike material. It is a soft, flexible, highly elastic material that can be custom formulated to perform in a variety of processes, including injection molding, extrusion, blow molding, and thermoforming. The material can easily be blended with other materials, including colorants.
Because TPEs contain no nitrosamines, phthalates, or PVC, they are good alternatives for medical tubing and other devices. TPE is a naturally translucent compound that is easily colored and bonds well to polypropylene. TPEs are being increasingly specified for medical products. This growing trend to replace latex and PVC is only one reason—the real strength of TPEs for medical applications is in their ease of manufacture and FDA compliance. Most are FDA CFR 177.12 10 compliant and are also approved for use in applications that require U.S. Pharmacopoeia (USP) XIX Class VI certification.4
TPE works well for antimicrobial blends used in molded parts with complex geometries, extruded profiles, tubing, thin film extrusions, extrusion blow-molded bags, injection blow-molded dropper bulbs, overmolded grips, pads, and seals. TPE remains clear when infused with antimicrobial coatings.
All TPE materials have the potential to be easily recyclable (i.e., green) because they can be molded, extruded, and reused like plastics, but they have the benefit of owning the typical elastic properties of rubbers, which are not recyclable owing to the thermosetting characteristics of rubber. TPEs also do not require added reinforcing agents, stabilizers, or cure systems. This characteristic leads to excellent consistency and also consumes less energy in production.
All medical material developers offer tailored plans for improving polymer properties. They also work with chemical providers to create not only innovative antimicrobial products and additives, but also to make sure there is a compatible match with the material that will flow through any tubing or come into contact with any device.
Another growing area is in the development of compatible colorants for medical tubing and devices. While clarity is important, color can play an important role too, particularly in the disposable container market such as with sharps containers. Also, specific materials flowing through tubing or catheters or materials that are held in containers may require UV protection while maintaining visual clarity. This is where specialized colorants are vital for many colored injected-molded antimicrobial medical devices
Application Examples
Partnering is the rule rather than the exception when dealing with the latest in both antimicrobials and tubing and fitting materials. The new breeds of antimicrobial tubing cover a wide range of applications.
New Materials Development. On the pure development end, companies involved in specialty compounds that tailor these to specific end-customer requirements are working with antimicrobial tubing facilities to test out the manufacturability of new compounds. Custom material formulations for both the tubing and the antimicrobial linings are first analyzed and tested for usefulness and compatibility, but then the actual determination of manufacturability of the products and true effectiveness of the antimicrobial formulation must be done by a cleanroom manufacturing facility.
Although all specialty antimicrobials and additives may perform in a lab, many simply cannot be made in real life. Therefore, a combination of initial material knowledge needs to be taken all the way through to manufacturing. One application may need fast-acting antimicrobials; another long lasting. Some need both.
It all comes down to the final product, how it will be used, and the type of performance the product needs. From the material development level, the range is very wide. It could be a wound-care device that may be on the body for eight hours or a military boot with a 10-year useful life—and everything in between. Although both need strict infection control in a clinical environment, a long-term IV system with tubing, bags, and connectors will have a different antimicrobial need from a single-use catheter. Therefore, it is important to be able to integrate what is needed into the end product while ensuring that the product is manufacturable.
Disposable Devices. A large market for silver ion antimicrobial linings is disposable devices in the urinary area: catheters, drainage tubes, drainage bags, and fluid-collection systems. Major companies are moving into stabilization of catheters, IV monitors, and IV lines. Sometimes it requires reinventing an old product in a new and better way. Other times it calls for a full paradigm shift with a completely new material.
The change from PVC to TPE tubing materials is being seen heavily in this disposables. To prevent urinary track infection requires using tubing that incorporates antimicrobials. Along with this materials change, several medical companies are also working with antimicrobial tubing providers on customized tubing materials to inhibit the ever-increasing biohazard risk of developing strains of bacteria and viruses that are simply immune to the antibiotics. Again, being a benign material, silver does not encourage the development of resistant virus or bacteria strains, so silver ion antimicrobials can address this before it can ever become an issue.
Ventilation Tubing. A recently FDA-approved endotracheal tube uses silver ion antimicrobial lining. This special tubing is intended for patients who must rely on a ventilator to breathe for 24 hours or more. Patients requiring such a breathing-support system are at risk of exposure to hospital-acquired bacteria that can build up on the breathing tube or pass through the tube to their lungs, eventually causing a lung infection known as ventilator-associated pneumonia (VAP). Bacterial pneumonia is one of the most insidious hospital-acquired conditions.
Luers and IV Connectors. Because IV assemblies are continually handled by clinicians, impregnating connectors on the inner and outer surfaces with silver ion prevents contamination and growth within the device of six pathogens known to cause catheter-related bloodstream infections. While clinics, hospitals, and other healthcare facilities follow highly sterile protocols, this is an added protection to reduce HAIs.
Consumer Pharmaceutical Devices. Pathogens can attack through various parts of the body, but the most common places for allergens, viruses, and bacteria to enter are through the nose and mouth.
Some companies are taking antimicrobial tubing out of the hospital and into the consumer market for items such as reusable catheters and, most recently, nasal irrigation (navage) systems for personal hygiene. Once out of the professional medical area of use, these products certainly benefit from the added safety of antimicrobial linings. One personal nasal hygiene product uses a two-bottle system with a micro diaphragm pump and a check valve that regulates the relative vacuum in the lower bottle. The vacuum draws saline from the upper bottle through the nose and then down into the lower bottle.
Antimicrobial tubing is also being used for the saline irrigation in 5/16 ID tubes. One company chose antimicrobial tubing with the silver ion interior lining because one of the tubes carries clean, pure saline, but when it comes out of the nose, it is not clean and it runs through the second tube to the bottle. In this case, the device manufacturer needed a non-PVC tubing with a specific durometer that was flexible, comfortable, and antimicrobial. A custom TPE formulation was developed only for this end application. Several different materials were evaluated before a specific type of flexible polyethylene was selected.
Conclusion
HAIs run billions of dollars annually in extended care and treatment, none of which is recouped by the medical facilities through insurance. Pathogens mutate quickly and render antibiotics useless in fighting them. Silver ion tube lining is part of the ammunition that the healthcare industry can use as one of the first lines of defense in this ongoing battle. It is effective across a broad range of bacteria and against mutating pathogens. It is also effective in blocking fungi and disease-forming yeasts, and it is highly effective against the formation of biofilms. While not the full solution to the war against HAI, it is definitely a superior weapon of defense.
Research for Tomorrow
The newest innovation for silver oxide antimicrobials involves surface-engineered ordered nanostructures of silver oxide that are built on the medical device surface. The approach employs nanotechnology to apply antimicrobial silver to medical devices. Some say that this nanotechnology may provide the most-effective platform to maximize the antimicrobial capability of silver in the future. However, currently there is no plug-and-play option. Customization is still the rule because there are so many different needs and parameters that must be addressed for each specific device or application.
Combinations of new materials such as the family of TPEs can enhance the ability of tubing to meet medical needs. Infection control is a rapidly growing concern. Antimicrobials used as coatings or integrated into materials are one avenue of attack against pathogens. Many uses for antimicrobial and material blends stretch beyond tubing and associated devices to medical and pharmaceutical needs including polymers, textiles, coatings, paper, and even adhesives. Constant innovation means a future of continual evolution in all medical device areas. Antimicrobial materials are a key part of this evolution.
References
1. Jeffrey Trogolo, “Protecting Devices with Silver-Based Antimicrobials, “ MD+DI, 32, no. 8: 52–58
2. “Antimicrobial Resistance in Healthcare Settings,” [online] (Atlanta: CDC, 2002); available from Internet: www.cdc.gov/ncidod/dhqp/ar.html.
3. “Study: Silver-Coated Catheters Reduce Urinary Tract Infections,” [online] (Mundelein, IL: Medline, 2008); available from Internet: www.medline.com/media-room/press-release.asp?ID=57.
4. “FDA Public Health Notification: PVC Devices Containing the Plasticizer DEHP,” [online] (Rockville, MD: FDA, 2002); available from Internet: www.fda.gov/MedicalDevices/Safety/AlertsandNotices/PublicHealthNotifications/ucm062182.htm.
Marcia Coulson is president of Eldon James Corp. (Loveland, CO).
About the Author(s)
You May Also Like





.png?width=300&auto=webp&quality=80&disable=upscale)