Punctal Plugs Take Control of Drug Release
The use of punctal plugs to treat dry eye patients isn’t new, but a University of Florida (Gainesville) prototype could offer a much-needed improvement.
January 1, 2010
|
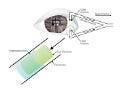
This punctal plug prototype both blocks tear drainage and delivers dry-eye medication. Image courtesy of UNIVERSITY OF FLORIDA |
“It is estimated that 10–30% of people suffer from the condition known as dry eyes,” according to a university press release. Usually caused by a lack of tears on the eye surface, the condition can lead to blindness.
Commercial punctal plugs treat dry eyes by reducing tear drainage from the eyes to the nose, which leads to an increase in tear volume. Punctal plugs developed at the University of Florida block tear drainage and also deliver dry-eye medication, thus providing dual treatment approaches from a single device. The reduced tear drainage improves the drug residence time in tears, resulting in increased efficacy and reduced side effects. The prototype also improves the drug-delivery process, enabling better control of release profiles than conventional plugs, says Anuj Chauhan, PhD, associate professor in the department of chemical engineering.
Chauhan’s plug design (see Figure 1) includes a drug-filled cylindrical core surrounded by an impermeable shell that covers about 50% of the core. The uncovered part is directed toward the eyes. To ensure a snug fit, the outer diameter of the shell matches the inner diameter of the canaliculus.
“The key difference between this design and all other designs is that the there is some space in between the uncovered core and the canaliculus wall, which will be filled with tears,” Chauhan says. “The drug from the core can diffuse radially out into this space and then diffuse axially out into the tears.” Conventional models lack this space, so the drug must diffuse from the circular cross-section of the plug that is in contact with the tears.
|
Figure 1. (click to enlarge) The design of the punctal plugs leaves space between the uncovered core and the canaliculus wall. |
This design modification creates a larger surface area for the drug to diffuse, thus offering better control of drug-release profiles. This characteristic makes it possible to obtain zero-order profiles without any initial burst. It also makes the release profiles less susceptible to reduced drug release caused by protein binding to the plug. According to Chauhan, another advantage is that the plug “has no burst release, which can arise in designs with shell covering the entire plug due to possible leakage from the space in between the shell and the core.”
The plugs are made from hydroxyl methyl methacrylate, a biocompatible material commonly found in ophthalmic applications, and the shell is made from silicone-based materials. The biocompatible material choice means that the plugs can be absorbed into a patient’s body instead of being removed.
Chauhan says that his prototypes currently release dry-eye drugs at a rate of about 3 μg/day, which is therapeutically effective for 45 days. Using an applicator, the plugs can be inserted into a patient’s upper punctum, lower punctum, or both. The permeability of the structure and the release duration can be customized per patient.
Several modifications are planned to further improve the device. Chauhan says that the single core will be replaced with multiple cores and may include “a third layer that surrounds the entire plug to minimize the drug transport into the walls of the canaliculus.” His research team is also exploring the delivery of other medication through the punctal plugs. Heng Zhu, a former student, and Chhavi Gupta, a current student of Chauhan’s, contributed to the invention. A patent is currently pending.
To license the technology, contact University of Florida’s Office of Technology Licensing at www.research.ufl.edu/otl/.
About the Author(s)
You May Also Like


.png?width=300&auto=webp&quality=80&disable=upscale)
