Could Exact Sciences Help Reverse This Troubling Cancer Trend?
March 2, 2017
With colorectal cancer on the rise in young adults, the maker of the Cologuard noninvasive home test for the disease says it might be time to adjust the screening age.
Nancy Crotti
Colorectal cancer is on the rise in young adults, according to a study by the American Cancer Society, which is recommending more screenings. That might represent an opportunity for a company that markets an alternative to the colonoscopy.
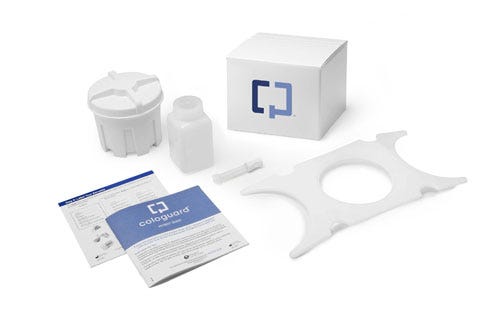
Exact Sciences (Madison, WI) makes Cologuard, a noninvasive home test for colorectal cancer, which FDA approved in 2014 for adults aged 50-85. Exact Sciences ships its Cologuard Collection Kit to the patient. The patient collects a stool sample in the kit and returns it to the company, which sends the result to the patient's doctor. Patients with positive Cologuard test results are advised to undergo a diagnostic colonoscopy.
Colorectal cancer primarily affects people age 50 and older. Among cancers that affect both men and women, it is the third most common cancer and the second leading cause of cancer-related death in the United States, according to the Centers for Disease Control and Prevention (CDC). The CDC estimates that if everyone age 50 or older had recommended regular screening tests, at least 60 percent of colorectal cancer deaths could be avoided.
The study, published in the Journal of the National Cancer Institute, recommends increased adherence to screenings among adults aged 50 to 54, as well as screenings for those under 50.
It found an increase in cancer of the distal colon, which is closer to the rectum, in those younger than 50, and of overall colorectal cancer rates among those 40-49. While middle-aged and older adults are more likely to have colorectal cancer, the study also revealed rising rates among those in their 20s and 30s.
The rates increased by 2.4% per year in those 20-29 and by 1.0% per year in adults age 30-39. In the mid-1990s, rates also began increasing in adults age 40 to 49 years (1.3% per year) and 50 to 54 years (0.5% per year). Rates declined among those 55 and older, which the authors attributed to increased screenings in that age group.
"Reversing increasing trends in adults age 50 to 54 years requires not only increased adherence to screening guidelines but also screening before age 50 years because the full benefit of polypectomy for preventing (colorectal cancer) requires about a decade to realize," the authors wrote.
Obtaining FDA approval to expand the age range for Cologuard to include younger adults would likely require more studies, time, and expense. But it has piqued Exact Sciences' interest.
"The point we're seeing from the study is there is an incredible need to look at adjusting the screening age and bringing more people into the screening population," said Exact Sciences spokesman J.P. Fielder. "The one thing we know about these long-developing cancers is, screening works."
Cologuard detects the presence of hemoglobin and biomarkers in DNA cells shed by large polyps as stool moves through the large intestine and rectum. In a clinical trial of 10,023 subjects, Cologuard detected 92% of colorectal cancers and 42% of advanced adenomas, while the fecal immunochemical test (FIT), a commonly used, non-invasive screening test that detects blood in the stool, detected 74% of cancers and 24% of advanced adenomas, according to FDA.
Most colorectal cancers start as abnormal raised or flat tissue on the wall of the large intestine or rectum. Some very large polyps, or advanced adenomas, and are more likely than smaller polyps to progress to cancer, according to FDA.
Nancy Crotti is a contributor to Qmed.
About the Author(s)
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
