Reshaping Devices with Microfeatures
Originally Published MDDI July 2006 MOLDINGAs the desire for small parts grows, manufacturers are designing medical devices with microsized features. This promising technology still has a long way to go.
MOLDING
|
(click to enlarge) Parts with features that measure in microns must meet tight tolerances for medical device use. |
Manufacturers are increasingly designing small devices such as hearing aids, ear canal implant devices, micropumps, and micromanipulators. The trend toward very small devices means that features on these devices need to become even more refined. Medical device manufacturers are designing products with microfeatures that require tolerances in microns (i.e., dimension variations that measure less than one-millionth of a meter).
The micromolding process is gaining momentum for producing plastic parts with such microfeatures. Injection molders have been producing these parts for a long time by optimizing their processes and equipment through trial and error. A device manufacturer looking to use micromolding needs to understand the benefits and limitations of the process and how best to use the technology.
For the most part, the concerns for micromolding are similar to those for conventional molding. It is important to have a supplier with technical knowledge, experience, and resources. However, to create a useful micromolded part, the manufacturer must also understand the unique aspects of the process, including mold building, processing, material handing, quality control, and part handling.
Mold Building
Precision mold building is key to the micromolding process. The first step in the process is to validate the mold steel. Mold build process validation is performed for two reasons. First, when making microscale features with micron-level tolerances, the mold steel must be extremely accurate, because there is no room for error when parts are microsized. If the mold steel is not right the first time, it must be rebuilt because most rework processes are not capable of the precision needed to modify the original mold steel. Second, many of the features in the mold are costly to measure because of the equipment and time required to inspect the microfeatures. In these cases, validation of the process may be less expensive than the verification of the mold to ensure steel conformance. In some cases, validation of the build process is just not possible, so steel verification is necessary. This increases the cost of the mold because rebuilding portions of the mold is the only recourse if the steel is not within required tolerances.
Microscale features with high aspect ratios require advanced mold-building techniques. These techniques include micromachining, micro EDM, microstereolithography, and laser ablation. LIGA techniques are also used to manufacture molds with high aspect rations. LIGA is a German acronym that stands for deep-etch lithography, electroforming, and molding.
It is critical to have EDM and milling machines with high positioning resolutions of 0.1 µm and accuracies of 1 µm to build molds that produce microparts.
Other aspects of mold building are not unique to microparts, although it may be more critical to ensure these aspects are designed well. Features such as undercuts and part ejection must be economical and feasible to construct.
With regard to economics, it is important to remember that micromolds do not adhere to common pricing models. That is, most people assume that as a mold gets smaller, the cost of material and labor should decrease. However, the equipment and methods required to create microfeatures and hold micron-level tolerances typically create an inverse relationship between mold cost and part size.
Because many of the mold-building processes employed for micromolds do not lend themselves to rework, the mold may need to be built several times to achieve the necessary steel tolerances. Additionally, the equipment necessary to hold the expected tolerances is on the leading edge of technology and, therefore, it is typically much more expensive.
Processing
The micromolding process requires machines with precise control over the metering and injection of the plastic melt. Injection capacity (the amount of time that the plastic material is exposed to the heating cylinder in the injection molding machine) increases as the part size becomes smaller. Shot weights of less than a gram are normal in micromolding.1
Recently, machine manufacturers responded to these requirements and built machines with electric servo drives to enable the machines to produce smaller injection units for more-precise process control. Some manufacturers downsized their injection units to minimize the residence time, while other manufacturers introduced an extruder and plunger concept. The minimized residence time improves the plastic melt quality, which is critical to the mechanical properties of the molded part. Questions that device engineers need to ask themselves before settling on the design features include:
• Does the material selected have the physical and mechanical properties required?
• What are the flow properties of the material when the wall thickness becomes very small (< 0.010 in.)?
• Does the supplier have knowledge and experience in molding the parts with microfeatures using selected materials?
• Does the supplier have the equipment needed to maintain process control in molding microparts?
An important factor to consider when selecting the equipment needed for micromolding is the resolution of the injection module. Taking shot-size resolution into account is critical for producing microparts. The molding machines used should be capable of controlling the movement of the injection piston or screw on the order of microns.
|
Table I. (click to enlarge) Typical shot-size resolutions in micromolding processes. OEMs should have the supplier try to obtain shot sizes smaller than those listed here (courtesy of Battenfeld). |
This capability is important because the piston or screw needs to be moved only a few microns to deliver the precise amount of plastic melt into the mold cavities. For better process control, manufacturers should use a shot-size resolution smaller than that required (see Table I). Exceeding shot-size resolution standards results in precise process control and consistent part molding.
Although the micromolding machinery is suitable for metal parts, limited information is available for processing of plastic materials. For example, flow properties are difficult to measure when wall thicknesses are in microns. An important flow property is the flow ratio, which is defined as the ratio between flow length and the wall thickness. One way to figure out the flow properties is to build a spiral mold.
In the injection molding industry, a spiral mold is used to determine the flow properties of a plastic material at certain wall thicknesses. The spiral mold can provide useful information about flow properties that is not available elsewhere. For example, a spiral mold built by one company suggests that flow ratios of 100:150 are common and suitable for manufacturing microsized products.2
Two different wall thicknesses, 0.150 and 0.225 mm, were considered to evaluate the flow properties. The material chosen was polypropylene with a melt index of 24, processed at the material manufacturer's recommended processing conditions.
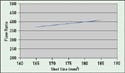
|
Figure 1.(click to enlarge) The effect of shot-size control on the flow ratio of a 0.15-mm-thick spiral. |
Figures 1 and 2 show the results of the experiment. Shot-size control has a significant effect on the plastic material flow properties in a micromolding process. In the conventional injection molding process, it is unlikely that a molder would discover how much shot-size resolution can affect process control because the molding machine does not have the accuracy needed to set the shot size required.
Materials
|
Figure 2. (click to enlarge) The effect of shot-size control on the flow ratio of a 0.225-mm-thick spiral. |
Material handling is another concern when it comes to micromolding. Medical device manufacturers often select exotic and expensive materials, such as bioabsorbable and implantable-grade plastics, for their components. These materials are expensive, partly because they are manufactured, tested, and packaged under cleanroom conditions. Conventional molds require a minimum of 1 g of material in the feed system to create a finished part that weighs only a milligram. Even though the shot-to-part weight ratio in micromolding is high, it is much lower than the shot-to-part weight ratio in conventional molding.
In medical device manufacturing, the sprue and runner are considered waste because OEMs do not allow regrind use. The usual practices of material drying to minimize the moisture content pose certain problems because the material required to mold microparts is minimal. Drying for longer periods might degrade the material and reduce the mechanical properties of the finished products. Manufacturers need to develop innovative ways of material handling and storage procedures to minimize or eliminate moisture absorption by the plastic materials.
Quality Control
Tight tolerances for plastic parts with microfeatures require advanced measurement technology. The resolution and accuracy of the measuring equipment should be able to capture the part-to-part variation within a micron. Vision-guided contact measurement systems are needed for accurate measurement of parts. Contact measurement systems with touch probe sizes in the order of microns are required for part inspection. Statistical process control, with the help of sensor technology, should be employed to minimize inspecting quality into the part.
Nanoindenting and atomic force microscopy inspection techniques are being evaluated to understand the characteristics of molded micro parts.4 The molding industry needs to accept these techniques and bring them into the production environment. In addition, standard test methods for different plastic materials need to be developed and adopted by the molding community.
Part Handling
Because micropart handling is an arduous task, it is essential that automation be integrated into a micromolding work cell. If the micropart is to become part of an assembly, automation at the manufacturing level eliminates the difficulty in locating and orienting the part downstream. Typical methods involve loading parts into custom fixturing or using tape and reel processes.
Conclusion
As product designers continue to miniaturize their parts, the boundaries of commercialized manufacturing practices will continue to be stretched. Scaling down existing technologies may provide some methods to produce parts with the desired features; however, long-term process capabilities require a more significant departure from conventional manufacturing practices. With the complex relationships between the mold, processing, materials, and part handling, manufacturers need to become experts in each of these areas.
Limited information exists for the manufacturers. Instead, they must invest in research and development to fully understand these interrelationships and also develop the manufacturing methods capable of cost-effectively producing microsized parts with micron-level tolerances. Process development using scientific methodology, validating the manufacturing processes, and maintaining the validation state using statistical process control techniques are the keys to successfully manufacturing parts with microfeatures.
Dana E. Schramm is director of engineering at Donatelle (New Brighton, MN). Raghu Vadlamudi is process development and technology manager at Donatelle.
References
1. Battenfeld micromolding literature (South Elgin IL: Battenfeld of America). Available from Internet at www.battenfeld.com.
2. J Johnson, R Panday, and R Vadlamudi, “An Investigation into the Effect of Process Variables in Micro Molding Processes,” in Proceedings of Molding 2006 Conference, (Las Vegas: Executive Conference Management, February 2006).
3. S Lee et al., “Ultrasonic Cavity Sensors for Micromolding,” in Proceedings of SPE ANTEC (Nashville, TN: Society of Plastics Engineers, May 2003).
4. BR Whiteside, MT Martin, and PD Coates, “Micromoulding-Process Characteristics and Product Properties,” in Proceedings of SPE ANTEC, (Nashville, TN: Society of Plastics Engineers, 2003).
Copyright ©2006 Medical Device & Diagnostic Industry
About the Author(s)
You May Also Like




.png?width=300&auto=webp&quality=80&disable=upscale)
