An effort to build a simulated model of the human heart shows how computer-aided engineering technology could revolutionize the way medical devices are developed, tested, and regulated.
June 2, 2015
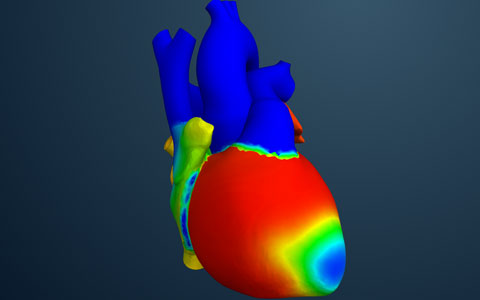
An effort to build a simulated model of the human heart shows how computer-aided engineering technology could revolutionize the way medical devices are developed, tested, and regulated.
Steve Levine
Dassault Systems's Living Heart Project aims to develop the first commercial-grade simulated model of the human heart. |
Computer Aided Engineering (CAE) technology has played a critical part in helping companies design and manufacture everything from jumbo jets to razor blades, enabling them to produce more efficient, cost effective, safe, and easy to use products and solutions.
Nowhere has this been more evident than in the automotive industry, where manufacturers have proven they can simulate the performance of new vehicle designs quickly and with low environmental impact. Using CAE technology, automakers can test products in the virtual world and select the best designs to bring into the real world. As a result, companies can now go from design directly to production with the confidence in the performance and safety of their product.
However, industries including healthcare have lagged in this transformation, in part due to the lack of accurate models, accepted processes and published standards covering CAE. As a result, the application of the technology has remained fragmented in research organizations scattered around the world. Now is the time to take the power of this technology and apply it to an objective that benefits us all: improved and affordable healthcare.
Start with the Heart
Cardiovascular disease is the No. 1 cause of death worldwide and costs the United States alone more than $400 billion annually. CAE technology can immediately begin helping to address challenges associated with testing new cardiac devices in humans. In fact, 95% of new devices in the United States are approved without ever being test in a human.
Imagine virtual human models that could test new medical devices to complement targeted human trials or be used to determine when a trial is unnecessary. Further, imagine having the ability to use data from a patient’s scans to develop a 3-D model of their beating heart. Prior to any invasive procedure, you could use the personalized heart model to simulate how the body will react to a stent or valve. Will the physician’s choice of valve be too big or too small? Is the procedure even necessary? Will the mechanical behavior of the heart be altered? How will it affect blood flow? What medications will best complement the procedure?
CAE can help answer those questions. The technology allows for truly individualized treatment and delivers greater insight into health and healthcare decisions.
The Living Heart Project
That is the goal of the Living Heart Project, a translational initiative launched by Dassault Systèmes in January 2014 that involves partnering with the medical community, researchers, manufacturers, and regulators to develop the first commercial-grade simulated model of the human heart.
Since it launched, the project has grown from three committed members to include more than 35 contributing organizations that have signed on to help build and validate the model. Never before has such a diverse group of experts had access to use and evaluate the same simulated model.
Further adding to the project’s momentum is the cooperation of FDA, which has signed a five-year research agreement that will initially target the development of testing paradigms for the insertion, placement, and performance of pacemaker leads and other cardiovascular devices used to treat heart disease. The hope is to not only fast-track the approval processes for new cardiac devices, but to inspire innovation within medical device design and manufacturing and provide patients with a greater sense of security and power when it comes to their own health.
Other Potential Applications
Imagine extending this approach even further, to orthopedics, the brain, or the eyes. We could one day simulate every system in the body, transforming the way patients are diagnosed and treated. We have the knowledge to do this, but do we have the will?
Imagine the benefits to humanity if we as a medical community continue to push these boundaries forward. We can use technology to not only disrupt the process of developing and testing medical devices and improve overall patient care, but to give individual patients more confidence in the treatments they receive.
Don't miss the MD&M East conference and exposition, June 9–11, 2015, in New York City. |
Steve Levine is chief strategy officer at Dassault Systèmes SIMULIA and director of the Living Heart Project.
You May Also Like


