13 Medtech Innovations to See at MD&M Minneapolis
August 22, 2016
The 13 entries for the MD&M Minneapolis Innovation Prize are but a sampling of what one can see on the expo floor of the largest medical device manufacturing event in the Midwest.
Dennis Engel and Chris Newmarker

From a vacuum-based decontamination mat to a super precise knife barrel hinge to mobile health-based breast cancer screening, there is a tremendous amount of wondrous variety among the 13 entries for the MD&M Minneapolis Innovation Prize.
The entries are a testament to the value of getting more than 600 medical device industry suppliers and more than 5000 industry professionals under one roof at MD&M Minneapolis, which runs September 21-22.
Check out the entries, and take our survey to help UBM editors select the five contest finalists to be included in a show floor tour.
|
(Image courtesy of Carl Zeiss Industrial Metrology) |
1. Computed Tomographs
Carl Zeiss Industrial Metrology (Maple Grove, MN)
Booth 1116
The Metrotom 800 and Metrotom 1500 industrial computed tomographs (CTs) offered by Carl Zeiss Industrial Metrology for component inspection during production are designed to work rapidly and have the flexibility to scan hybrid workpieces made of various materials. The former CT is available in a 225-kV, 500-W version as well as the original version whose tube voltage is 130 kV and power output 39 W; the extended-performance 225-kV machine provides faster scanning over a wider range of applications. The latter CT, a new development, has a measuring z-axis that enables components as large as 700 mm in height to be thoroughly scanned. It allows the workpiece to be raised and lowered by 400 mm and enables multiple scans to be merged into one volumetric model. Associated visualization and evaluation software delivers 3-D images and section views based on voxel data generated by a proprietary operating system, and a specialized software system optimizes performance of metrological evaluations. The interior measuring space is always completely in view, either from a vantage in front of the machine or from a separate computer workstation.
|
(Image courtesy of Circuit Solutions) |
2. Decontamination Floor Mat
Circuit Solutions (Minnetonka, MN)
Booth 1340
The Vortex Air WalkMat from Circuit Solutions uses vacuum technology to clean equipment wheels and the bottom of all types of footwear before and after people enter critical spaces in hospitals, clinics, and operating rooms, as well as cleanrooms and air showers in device manufacturing facilities. Registering the weight of arriving individuals or equipment, retractable balls on the surface of the mat activate the vacuum to remove particles, hair, and debris that might be tracked into a controlled-environment area. The manufacturer attests to the mat's ability to remove 90% of 1-to 5-µm particles from clean spaces within one day of installation and up to 90% of submicron particles within a few days. Its vacuum system returns HEPA-filtered air to the area. Whereas conventional methods of debris and hair removal rely on sticky paper floor mats that have to be replaced regularly and whose reliability is often questionable, this HEPA-centered system, once installed, can be counted on to minimize contamination and optimize patient outcomes with little further effort in the matter of clean-space entry.
3. 3-D Printed Rapid-Prototyping Molds
Diversified Plastics (Minneapolis)
Booth 2138
The injection molding specialist Diversified Plastics is able to use 3-D printing to create injection molds for producing prototypes. By means of this process, the company can mold prototype parts, using the actual resin selected for production, more quickly and more economically than if soft-steel or aluminum molds were constructed and employed. In addition, prototypes made from the 3-D-printed molds can be subjected to functional and sterilization tests. The upshot is that molding prototype parts in 3-D-printed injection molds is a way to minimize product development times and costs; in one case, the molder used the method to help a urology device manufacturer cut prototype development costs by a reported 88% and turnaround time from five weeks to one week. A pioneer in the construction of prototyping molds via 3-D printing, the company has strived to remain on the leading edge of the technology.
|
(Image courtesy of Diversified Plastics) |
|
(Image courtesy of Keyence Corp. of America) |
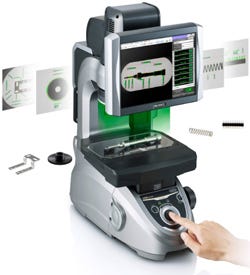
4. Visual Inspection System
Keyence Corp. of America (Itasca, IL)
Booth 1811
Offering high speed and accuracy of ±2 µm, the IM-6225 Instant Measurement system is able to inspect at the push of a button as many as 100 medical device parts placed on a glass stage in any orientation. Manufactured by Keyence Corp. of America and intended to substitute for various conventional 2-D-measurement tools, the visual inspection system can identify a part, such as an orthopedic screw, tube, syringe, or vial, pull up the program designed for measuring that part, and measure up to 100 surface dimensions on the part, automatically adjusting the lighting and focus and repositioning the program to suit each part on the stage in turn. It recognizes the profile and reflective images of each part, compares them to preprogrammed tolerances, produces a pass or fail record for each dimension, and saves the data locally or to a network. Another button press produces a full printed inspection report, and all of this is completed in 3 seconds. The system uses 24 internal lenses, four of which are 4-in. telecentric lenses that provide a 40-in. field of view. It uses preloaded macros to handle complex and repetitive shapes, provides a simple and intuitive user interface, and is portable between lab and shop floor without requiring recalibration.
5. A Super Precise Knife Barrel Hinge
Liquidmetal Technologies (Rancho Santa Margarita, CA)
Booth 2432
The Liquidmetal Hybrid Knife, which launched in fall 2015, includes a patent-pending barrel hinge that reduced part count for the knife to two, versus the multiple components typically required of barrel hinges. The transition between the open and closed position of the blade and its protector is at a smooth and precise 12.7 µm. Says Liquidmetal: "There are a broad range of medical device applications that could benefit from the barrel hinge, coupled with the design flexibility, precision, and material properties of Liquidmetal alloy. Many endoscopic devices today rely on opening and closing end-effectors whether it be for suturing, cutting, clamping, gripping, or sealing tissue."
|
(Image courtesy of Liquidmetal Technologies) |
|
(Image courtesy of Marshall Manufacturing Co.) |
6. Automated Wire Bending
Marshall Manufacturing Co. (Minneapolis)
Booth 2048
A specialist in custom processing of medical device components made from tubing and wire, Marshall Manufacturing Co. has been able to virtually eliminate traditional manual bending from its repertoire in favor of operations involving highly technical automated and robotic equipment. The company has concentrated on perfecting its capabilities in bending premachined wire and tubing in order to help medical OEMs fulfill challenging design goals and requirements. When a device requires custom bending of wire or tubing, with a specified component orientation at the tip or end or within the bend to enable particular targets within the body to be reached, the contract manufacturer can bring to bear specialized equipment, custom software and tooling, and in-house process design engineering capacity. It counts among its achievements the development of a solution to the problem of precise tolerances in machined component features being lost when premachined wire or tube is subjected to bending. Its system holds those tolerances throughout the bending process.
7. Optical Encoders
Micromo (Clearwater, FL)
Booth 1249
|
(Image courtesy of Micromo) |
Micromo in July launched its Faulhaber IER3 optical encoders, which are able to position Faulhaber motors with a high degree of precision, within 0.1° to 0.3°, as well as repeatability, typically only 0.05°. Besides the accuracy and repeatability, the Faulhaber IER3 optical encoders are also resistant to strong magnetic interferences. Such resistance makes them especially attractive for applications with magnetic interferences including tomography and medical image scanning. Another plus is that the the LED, photodetectors, analysis unit, and interpolation levels are installed in one chip, so the encoders take up minimal space. According to Micromo, the encoders boast a resolution of up to 10,000 lines per revolution. They have an angular resolution of 0.009°, with the evaluation of 40,000 edges per revolution.
8. A Mobile Health Breast Cancer Screening Solution
Pressure Profile Systems (Los Angeles)
Booth 2538
The SureTouch system is a cloud-based medical device that includes a wireless handheld touch sensor, touch screen display, and a clinical information system to support telemedical consultations. The SureTouch's proprietary tactile sensor technology creates a high-resolution, handheld pressure sensor array that together with processing algorithms is able to identify masses in breast tissue. Says Pressure Profile Systems: "This digital, enhanced sense of 'touch,' allows SureTouch to dramatically improve upon the limit of lesion size and depth detectable by conventional manual palpation techniques. ... The portability of the device combined with the ability of a nurse to perform exams without needing a physician present at the exam site allows screening to truly be delivered into communities, removing the common barriers of inconvenience and access."
What is SureTouch Mobile from ELI YERED on Vimeo.
|
(Image courtesy of ProPlate) |
9. An Improved Way to Coat Plastic With Metal
ProPlate (Anoka, MN)
Booth 2523
ProPlate's Meta-Poly coating innovation hit the market in mid-2016. The proprietary surface preparation and custom plating process is able to coat a layer of metal onto plastic, without the use of chemical etching or conductive paints. ProPlate officials think medical device applications are vast for Meta-Poly, but it could be especially useful for catheter applications, providing a better way to add radiopaque markers and current conducting paths to polymers. Because Meta-Poly can apply metal thinner than traditional methods, it can impart a performance advantage of a lower profile while reducing material costs.
10. Online DFM Analysis
Proto Labs (Maple Plain, MN)
Booth 525
ProtoQuote is an automated interactive quoting system offered by Proto Labs through its Web site, whereby medical device engineers can upload their 3-D CAD models and receive free design for manufacturability (DFM) analysis and real-time pricing information within hours. By means of proprietary software and compute clusters, the system analyzes the CAD model to generate an interactive price quote and design analysis, with very little human interaction being necessary. The software has been continually enhanced; the current version can offer a highly detailed design analysis that includes an interactive three-dimensional image that enables developers to review their part geometry and assess any potential design issues such as sink or internal undercuts, which are highlighted. The system allows developers to iterate, obtain premarket approvals, pass clinical trials, and launch products as quickly as possible. The service provider, which originated this type of software, released its current version of the quoting system in 2015.
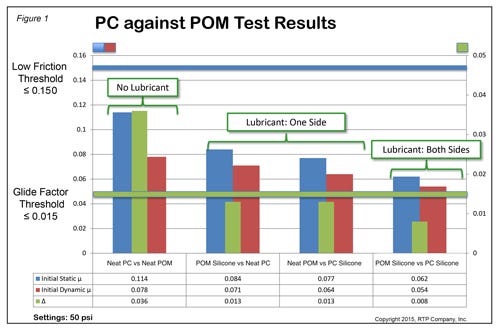
11. Plastic-Against-Plastic Friction Database
RTP Co. (Winona, MN)
Booth 1711
RTP Co. says it has tribological database like no other for medical device designers. Data comes from the plastic-against-plastic friction test RTP developed in 2014. Measurement is based on a core data point known as Glide Factor SM. Device designers are able to screen plastic-on-plastic friction pairs in order to achieve safer and more effective material selections for devices including injection pens, auto-injectors, stop cocks, inhalers, and safety syringes. Says RTP: "Optimal low friction, one-time performance depends on reducing the system friction in two critical measurements: the lowest possible static coefficient of friction and the lowest possible Glide Factor SM. Using the test data collected in the tribological database, a designer can make proper material selections for these devices and reduce or eliminate the stiction phenomenon. ... Understanding Glide FactorSM and predicting plastic-on-plastic behavior can be highly beneficial to device manufacturers and ultimately, the patients."
|
(Image courtesy of RTP Co.) |
|
(Image courtesy of Teleflex Medical OEM) |
12. Ultrastrong, Ultrathin Orthopedic Tape
Teleflex Medical OEM (Gurnee, IL)
Booth 2425
A specialty product developed by Teleflex Medical OEM is the Force Fiber OrthoTape Braid, an ultrastrong, ultrathin tape whose low profile and broad footprint makes it suitable for surgical applications in which tissue pull-through must be prevented. It is constructed of durable, pliable ultra-high-molecular-weight polyethylene and is stronger than round suture of similar size and material. Its one-piece braiding configuration makes this tape thinner than other tapes. In addition, tests have shown it to be more pliable. With uses that include fixing tendons or ligaments to bone and stabilizing joint dislocations, the tape is designed for optimum pressure distribution, and its uniform compression helps prevent strangulation of tissue or the cutting of tissue and bones that may occur with round sutures. It is cleared in the United States for approximation or ligation of soft tissue, including the use of allograft tissue for orthopedic procedures. Available in various colors, patterns, and widths between 1 and 5 mm, the tape can be customized to suit the application.
|
(Image courtesy of Zeus) |
13. Non-Metallic Braid Reinforcement for Vascular Catheters
Zeus (Orangeburg, SC)
Booth 2105
The Zeus LCP monofilament fiber, which Zeus is launching at MD&M Minneapolis, boasts the performance of stainless steel wire braiding used in catheters for vascular procedures--without actually using metal. Think MRI compatibility, which is increasingly important. That's because better visualization of soft tissue is needed more than ever as minimally invasive medical procedures increase in complexity. The LCP monofilament is braided over a catheter base liner during construction to provide mechanical strength and support, with the braiding then covered with a layer of Pebax or another nylon jacketing material reflowed under heat shrink. The reflow and bonding process causes the braiding to become fused between the nylon bonding material and underlying liner, achieving structural reinforcement. Zeus's proprietary processes are able to create a fiber with extremely low potential for fraying. Zeus says the Zeus LCP monofilament-braided catheters "retain torquability and stiffness while still allowing deflectability at the distal end of the device."
Create your own user feedback survey
Chris Newmarker is senior editor of Qmed. Follow him on Twitter at @newmarker.
Like what you're reading? Subscribe to our daily e-newsletter.
About the Author(s)
You May Also Like










.png?width=300&auto=webp&quality=80&disable=upscale)
