Catheter and Stent Fabrication 15578
Wireless data collected directly from patients during normal physical activities could enable the development of better knee implants
September 5, 2007
Originally Published MPMN September 2007
SPOTLIGHT
Catheter and Stent Fabrication
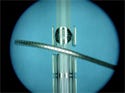
Catheter-tipping system
|
A company’s catheter-tipping system is suitable for tipping and welding almost all thermoplastic materials. It features programmable controls, a stepping motor drive for precision feeding, and quick-change tool design. Manufactured for industrial use, the machine is powered by a 1-kW at 450-kHz radio-frequency generator. Machining capabilities include round or irregular tips, multilumen shape transitions, soft-tip fusion, neck-downs, butt welds, angular welds, and balloon-to-catheter welds.
Cath-Tip Inc., Sandy, UT
www.cathtip.com
Catheter manufacturing
|
Operating in a Class 100,000 cleanroom, a company produces molded and extruded catheter components in standard and custom configurations. Radio-frequency technology is used for catheter tip forming, including soft-tip forming. Catheter drilling and printing are also available. The company offers research and development, design, and engineering services, as well as manufacturing of subassemblies or finished products.
Martech Medical, Harleysville, PA
www.martechmedical.com
Laser tube cutting
|
Laser tube-cutting equipment is suitable for medical implants made from materials such as nitinol, stainless steel, and cobalt-chromium alloys. Potential applications for the equipment are vascular stents, vascular closures and connectors, and catheter-based deployment components. The technology also provides the capability to measure the same feature on a precision-cut tube repeatedly to within 50 millionths of an inch.
Laserage Technology Corp., Waukegan, IL
www.laserage.com
Fastening station
|
A fastening station orbitally forms parts up to 0.312 in. in diameter or an equivalent area in tube, square, or other format. Typical cycle time for the B-310 is less than 3 seconds for materials such as aluminum, brass, bronze, copper, lead, zinc, stainless steel, and composite plastics. Suitable for high-volume applications, the product has an adjustable tool stroke from 0.06 to 1.75 in. within increments of 0.001 in. Standard throat depth is 6 in., and the pneumatic orbital riveter provides more than 1 tn of force on a 5/16-in. head.
The Orbitform Group, Jackson, MI
www.orbitform.com
Catheter-tip-forming dies
|
Manufactured using the company’s nickel-cobalt alloy, catheter-tip-forming dies are electroformed on CNC-machined mandrels. NiColoy dies possess electromagnetic, thermal, strength, and corrosion characteristics closely matching or exceeding those of stainless steel, according to the company. Multiple dies can be formed on the same reusable mandrel. Dies can be supplied with hubs for use in radio-frequency induction, hot-air, and other types of tip formers.
NiCoForm Inc., Rochester, NY
www.nicoform.com
Stent tubing
|
Designed specifically for the requirements of stents, a company’s implant-grade tubing is seamless and possesses suitable concentricity. Materials include 316LVM, nitinol, L605, MP35N, 35NLT, Elgiloy or phynox, tantalum, and special alloys. The 35NLT is a grade of cobalt alloy elaborated by VIM-VAR practice, and is designed to bolster material fatigue life. Nitinol tubing at the deployed stent size is also available.
MiniTubes, Grenoble, France
www.minitubes.com
Laser catheter bonder
|
An alternative to such bonding methods as adhesives, hot-air, or radio-frequency, a laser catheter bonder is designed for proximal and distal balloon bonding, soft-tip attachment, and shaft bonding. The system features automatic sample clamping, an integrated LCD, and a programming interface. No programming experience is necessary, according to the company. Mechanically, the system features dual precision spindles, a large through-bore for long sample loading on both sides, spindle speed adjustment, and soft-touch clamping to protect the samples. The laser provides adjustments for power, spot size, and focal point.
Automation GT, Escondido, CA
www.automationgt.com
Copyright ©2007 Medical Product Manufacturing News
You May Also Like