These Device Startups Are Challenging the Status Quo
Medical device startups are taking on established technologies in a quest to change the standard of care.
March 1, 2016

Arundhati Parmar
In the world of healthcare, disrupting the standard of care means getting payers, providers, and even patients to abandon what they know for something new. It’s a herculean task that many medical device companies shy away from, preferring instead to focus on incremental innovation.
Still, some companies rise to the challenge—and often they’re not industry titans but instead scrappy startups.
Here are three upstart medtech companies out to upset the apple cart with technologies that take on the current paradigm of care.
The companies profiled below have taken on this endeavor because executives passionately believe that their way expands the choices available to patients, is more clinically effective than the entrenched care paradigm and perhaps, most importantly, is more cost effective.
|
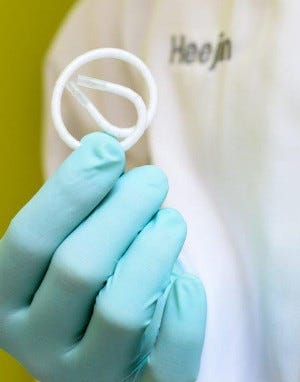
Taris Biomedical's device for treating OAB |
Lexington, MA-based Taris Biomedical wants to reshape the treatment for overactive bladder and bladder cancer, among other urological diseases, with the help of a novel drug-delivery system.
The company’s CEO contends that the reason most drugs are ineffective in treating overactive bladder (OAB) for some patients is that the drug is delivered orally, far from the source of where the problem lies. For patients for whom daily life has become a burden because of worsening OAB symptoms, urologists have come up with a work around.
“The best a urologist can do today is . . . just squirt a solution of the drug in the bladder through a catheter,” said Taris Biomedical CEO Purnanand Sarma. “It’s not uncommon in Europe for women with severe incontinence to grind up Detrol tablets [and] catheterize themselves six times a day.”
But this method is not effective because the bladder simply expels the drug a short time later when the patient has to urinate.
“The standard of care today is squirting a solution into the bladder and hoping the patient can keep it,” Sarma quipped.
Patients whose quality of life has been severely compromised by OAB often take the drastic decision of undergoing Botox treatment or surgery to live with an implanted device such as Medtronic’s InterStim, which treats OAB by disrupting electrical signals from the brain to the nerves that control the bladder.
But Sarma believes a drug-delivery device Taris Biomedical is developing could be a a more convenient office procedure for patients for whom oral drug-delivery has failed.
Based on intellectual property that came out of Robert Langer’s lab at the Massachusetts Institute of Technology, the Taris device is essentially a silicone tube comprising two lumens. One lumen contains a solid drug core and the other features a wire form made of nickel-titanium alloy to impart shape. The device can be introduced in a linear fashion by a conventional catheter, but it then contorts into a pretzel-like shape to prevent expulsion by the bladder.
Medtech entrepreneurs: Enter your startup in MD+DI's Medtech Startup Showdown! |
When the bladder is filled with urine, the liquid covers the entire length of the semipermeable silicone tube and enters the drug-filled lumen. The drug dissolves, and osmotic pressure builds up. The drug is then released into the body through tiny laser drill holes that regulate how much of the drug is delivered.
Sarma believes that because the drug is delivered by a device contained locally in the bladder, Taris’s product can actually release the drug in much lower doses than through conventional OAB treatment.
“It is not just sustained release; it is local sustained release,” he said. “That’s because the dose that we give is orders of magnitude lower than what you give orally.”
And the Taris solution isn’t limited to OAB treatment. The company has also shown that using Lidocaine can help shrink bladder cancer lesions if it is administered using the local sustained delivery. That’s a markedly different and more effective approach than cauterizing bladder cancer lesions under general anesthesia, Sarma contends.
The Taris drug delivery system is currently in trial phase. The company plans to engage in four or five clinical trials over the next 15 months to get FDA clearance for each of the different indications it is seeking.
Sarma said he is confident of the clinical efficacy of the sustained local delivery model. Taris sold a similar concept for treating interstitial cystitis to Allergan in 2014 for $587.5 million. Recently, Taris raised $32 million to help advance its local sustained delivery platform and transform itself into a urology company.
|
The Zio XT Patch from iRhythm Technologies |
With a product already on the market, iRhythm Technologies is further along in its journey to change the care paradigm than Taris Biomedical. The San Francisco-based startup is aiming to disrupt the way cardiac arrhythmia is diagnosed. .
Typically, cardiologists suspecting a condition such as atrial fibrillation send patients home with a holter monitor, which records a patient’s EKG continuously for two days. With wires and cables, the method—thegold standard for atrial fibrillation detection—is cumbersome, to say the least. The National Heart, Lung, and Blood Institute (NHLBI), advises patients using a holter monitor to avoid using electric blankets and toothbrushes and forgo showers to prevent the possibility of electrical interference.
“You can take a sponge bath instead,” the NHLBI advises.
iRhythm’s Zio XT Patch offers an alternative. Worn on the chest, over the heart region, the device is a single-use, water-resistant, continuous cardiac rhythm monitor that can be worn for up to 14 days.
During this period, patients can go about their daily routine while the patch collects heart rhythm information and sends it to iRhythm, which gathers and analyzes the information and issues a report at the end of the monitoring period. Patients can also press a button on the patch if they are experiencing symptoms associated with arrhythmia.
As of November, the Zio XT Patch had been prescribed to almost 400,000 patients. Kevin King, the iRhythm’s CEO, explained that the company has successfully expanded payer coverage for the device to 290 million patients.
|
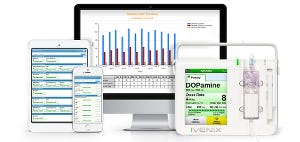
The Ivenix Infusion Management System |
Amesbury, MA-based Ivenix is engaged in a true David-versus-Goliath battle. This early-stage startup is just beginning to test and validate a radically new infusion pump system it hopes will take out the competition.
“This is a zero-sum game, and we would like to replace some of the existing participants in the pump world,” said Ivenix CEO Stu Randle.
The existing participants he’s referring to include Hospira, Baxter International, and CareFusion, (now part of Becton Dickinson), and all of them have run afoul of FDA due to malfunctioning pumps and software over the past several years.
In fact, between 2005 and 2009 FDA received approximately 56,000 reports of adverse events associated with the use of infusion pumps, including numerous injuries and deaths. In this period alone, 87 recalls occurred. In 2010, FDA undertook the Pump Improvement Initiative to reduce medication errors tied to pumps and exhorted pump makers to improve the design of these devices.
To Ivenix, formerly called Fluidnet Corp., this smelled like opportunity. The company is developing the Ivenix Infusion Management System, which it calls an “intelligent” infusion pump system, to improve nursing efficiency, improve the software system that current pumps have, and better integrate with the hospital’s pharmacy and electronic medical record (EMR).
For ease of use, the Ivenix pump’s screen and interface are inspired by smartphones. On the pump side, Randle said the technology is geared to improve patient safety and increase nurses’ workflow efficiency.
“We use a fundamentally different pump technology, which is pneumatic, and the value there is that it allows us to deliver incredible accuracy over a very long period of time across a wide range of flow rates, viscosity and other things,” Randle explained. “Most pumps today, the flow rate can vary by the extent to how high the bag is or how low, is it kinked or not kinked. So we actually constantly measure the flow rate and adjust.”
The Ivenix Infusion Management System’s software is also an improvement over that of traditional infusion systems. It allows nurses to see the patient’s medication order straight from the pump instead of having to pull it up on a separate screen tied to the hospital EMR. The system integrates with the hospitals’ pharmacy side to show whether those medications are available, and, it can automatically program itself based on orders or instructions from a barcode medication administration system.
“If you are outside of the range of those instructions, the pump won’t let the nurse start [the drug delivery],” Randle explained.
Further, the pump has drug compatibility software built in, as well as the ability to integrate with each patient’s EMR, so nurses won’t need to manually record patient infusion data in their EMRs.
All of this improves the workflow and efficiency of a busy nurse managing multiple patients in the emergency room or intensive care unit. It also reduces the risk of medication errors, which could boost patient safety and help hospitals keep costs down, Randle noted.
Randle believes the Ivenix system can actually bring in revenue, too.
“It’s our belief and understanding that hospitals miss a lot in the charging for delivery of fluids—particularly in the ER—and we believe, because of our IT connection, that we can improve that,” he declared.
Ivenix has to prove all of its claims, of course, and testing and validation has begun. The company expects to file a 510(k) submission with FDA at the end of the year. In August of 2015, Ivenix closed a $42 million round of funding. After that, Randle, the former CEO of GI Dynamics, was appointed to the company’s chief executive spot.
“These guys have really accomplished a ton in the last two years and really brought the product through to [having] the potential to really change infusion therapy in the hospital,” he said just three weeks after his appointment.
You May Also Like


.png?width=300&auto=webp&quality=80&disable=upscale)
