Medtech Startup Showdown 2016: Round 1—Noninvasix vs Advanced Prenatal Therapeutics
Noninvasixvs.Advanced Prenatal Therapeutics
March 28, 2016
| vs. | ||
|
|
|
|
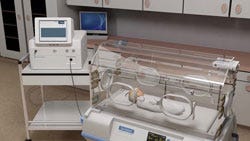
Describe your device and how it will benefit healthcare. | Each year, 270,000 preterm and low-birth-weight babies are born into a life of brain impairment due to a lack of blood flow to the brain after birth. Called hypoxic ischemic encephalopathy, this condition is a precursor to cerebral palsy and is responsible for 23% of all neonatal deaths and costs an average of $28,000 per patient. There is currently no technique for easily, repeatedly, and noninvasively monitoring or measuring cerebral circulatory adequacy. Using patented optoacoustic technology, Noninvasix is developing a monitoring system for at-risk babies in the NICU to measure the amount of oxygen in the brain in real time. Using a sensitive, wide-band head strap to access the superior sagittal sinus through the front and back fontanelles of the baby’s still-forming skull, Noninvasix’s optoacoustic monitoring system sends short laser pulses of near-infrared light into the brain. Hemoglobin in the blood absorbs the light at different frequencies depending on whether or not it is carrying oxygen. Absorption causes rapid thermal expansion of the hemoglobin resulting in a measureable acoustic wave whose amplitude is proportional to the concentration of hemoglobin. An acoustic transducer then detects these waves, and time resolution of the resulting signal determines the depth from which the signal was derived. The high resolution of this optoacoustic technique has been shown in pre-clinical studies to yield real-time and continuous measurement of blood oxygenation. |
| Advanced Prenatal Therapeutics’ Targeted Apheresis Column for Preeclampsia (TAC-PE) is the first medical device developed specifically for the treatment of preeclampsia. Preeclampsia affects 5-8% of all pregnancies in the United States and is a leading cause of maternal death, fetal death, and prematurity. The TAC-PE consists of a medical-grade plastic column containing proprietary targeting agents that bind to certain disease-causing factors that are present in the patient’s circulation. These abnormally-elevated factors are filtered from the patient’s blood as it is pumped through the TAC-PE using existing apheresis machines. Treatment with the TAC-PE is anticipated to alleviate life-threatening symptoms of preeclampsia and to delay premature birth by several weeks. Extending the time the fetus has to develop inside the womb will not only dramatically decrease neonatal intensive care (NICU) costs, but also profoundly improve fetal outcomes (e.g., reduced incidence or severity of neurological damage, lung disease, deafness/blindness, learning disabilities, cerebral palsy, etc.), which will have a profound life-long impact on healthcare systems and society. |
How does your product differ from the competition? | In contrast to other purely spectroscopic techniques, such as near-infrared spectroscopy, this technology provides an absolute, rather than relative measurement, and can be targeted to specific blood vessels, such as the sagittal sinus vein in the brain. Such capabilities allow optoacoustic oximeters to be more broadly applicable than pulse oximetry. Furthermore, all NIR spectroscopic techniques rely on returning or transmitted light; therefore, they have limited ability to separate the signal derived from venous saturation, which reflects tissue oxygen uptake, and arterial saturation, which represents a component of oxygen supply. Although NIRS techniques have provided vital information on neonatal cerebral circulation they have not yet proven useful for routine clinical monitoring. Real-time monitoring is not a reliable feature of the small catheters currently utilized in the neonatal and pediatric setting. |
| Currently, preeclampsia is managed with bed rest, close monitoring, and administration of certain drugs that alleviate life-threatening symptoms (e.g., high blood pressure and seizures). Due to their toxicity to the unborn baby, these drugs are typically only used to stabilize the mother long enough to induce premature delivery (i.e., for two to seven days only). The TAC-PE treats life-threatening symptoms of preeclampsia by physical removal of the agents associated with disease progression and without the use of toxic drugs. Additionally, where current treatments focus solely on the survival of the mother, the TAC-PE enables pregnancy to be prolonged (i.e., possibly by two to three weeks or more), resulting in substantially improved fetal survival and mitigation of congenital defects. |
Do you have customers yet? | A third-generation prototype has been safely tested in adult humans and neonates in Houston, TX. |
| Advanced Prenatal Therapeutics is in the preclinical stage of development. The commercial prototype has been fully designed and non-clinical testing is expected to be substantially completed in 2016. Partners for clinical testing have already been identified in anticipation of clinical studies in early 2017. |
How much money have you raised? | $2.2 million |
| Approximately $285,000. This has supported primary design/development, implementation of an ISO-certified quality system, patent allowance of the key IP, and early-stage non-clinical testing. |
Who are your investors? | University of Texas Medical Branch, the Texas Emerging Technology Fund, and private investors |
| Friends/family and angel investors |
What is the next milestone for your device? | A Series A round in the amount of $3.75 million is underway. Within 24 months of closing the Series A, Noninvasix expects to complete a production prototype, perform clinical trials and obtain FDA 510(k) clearance. The company submitted a presub to FDA in February 2016, with expectations of a decision on the appropriate clinical regulatory pathway in April. |
| The current goals for 2016 are to finalize the commercial prototype and complete all nonclinical testing to enable pivotal clinical studies beginning in 2017. |
You May Also Like




.png?width=300&auto=webp&quality=80&disable=upscale)
