Intuitive Stock Surges 13% As It Readies New Surgical Robot
April 1, 2014
Intuitive Surgical has announced FDA clearance and the U.S. launch of its new da Vinci Xi Surgical System.The company, in a Tuesday news release, described the Xi as a "technological leap forward in replacing large-incision abdominal surgeries (open surgery) with a minimally invasive approach." The company plans to market the da Vinci Xi System around the world.News of U.S. approval of its latest robotic-assisted minimally invasive surgery system caused Intuitive's stock to rise nearly 13% in value, to nearly $494 per share, at the close of trading Tuesday. It's now up more than 28% year-to-date."The da Vinci Xi System's new overhead architecture means that multi-quadrant surgery can be performed without repositioning the system, an innovation long sought by surgeons who perform complex procedures," Gary Guthart, Intuitive Surgical's president and CEO, said in a news release.Intuitive needs the boost, because Guthart and other executives in the company have been engaged in damage control in the discourse regarding the safety of Da Vinci robotic surgery. Guthart has complained that despite reported adverse events and ongoing FDA issues, surgery with the Da Vinci system is still far safer than conventional open surgery.Medgadget is also reporting on new competition for Intuitive Surgical from SOFAR, an Italian pharmaceutical firm, that has its own surgical robotics platform: "The Telelap ALF-X features haptic feedback, allowing the surgeon to indirectly 'feel' the tissues that are being manipulated. This can lead to improved safety and allow certain maneuvers to be performed with greater confidence."
|
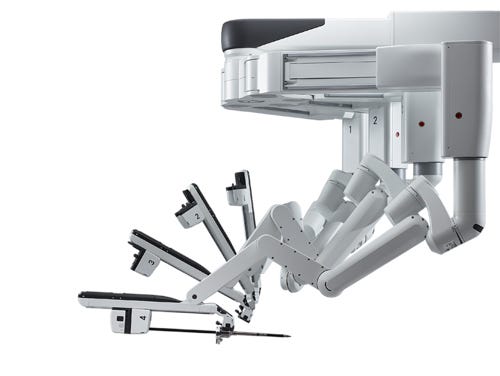
The arms of Intuitive Surgical's da Vinci Xi Surgical System, as shown on its website. |
When it comes to its stock price, Intuitive Surgical is one of the best performing medical device companies of early 2014. But it is also worth noting that it was trading at more than $500 a share a year ago. The da Vinci Xi System expands upon core da Vinci System features, including wristed instruments, 3D-HD visualization, intuitive motion, and an ergonomic design. Improvements upon older models include a new overhead instrument arm architecture designed to facilitate access to a patient from virtually any position; a new endoscope digital architecture that creates a simpler, more compact design; an ability to attach the endoscope to any arm, improving visualization; and smaller, thinner arms with newly designed joints to improve range of motion.
Refresh your medical device industry knowledge at MD&M East, June 9-12, 2014 in New York City. |
Intuitive Surgical also plans to seek regulatory clearance to make the da Vinci Xi System compatible with Intuitive Surgical's Firefly Fluorescence Imaging System.
Related Content
5 of the Best Performing Medical Device Companies of Early 2014
Chris Newmarker is senior editor of MPMN and Qmed. Follow him on Twitter at @newmarker.
About the Author(s)
You May Also Like