Could a New Catheter-Delivered Implant Satisfy Unmet Patient Needs?
August 1, 2017
A new implant size aims to meet the needs of a wider patient population.
|
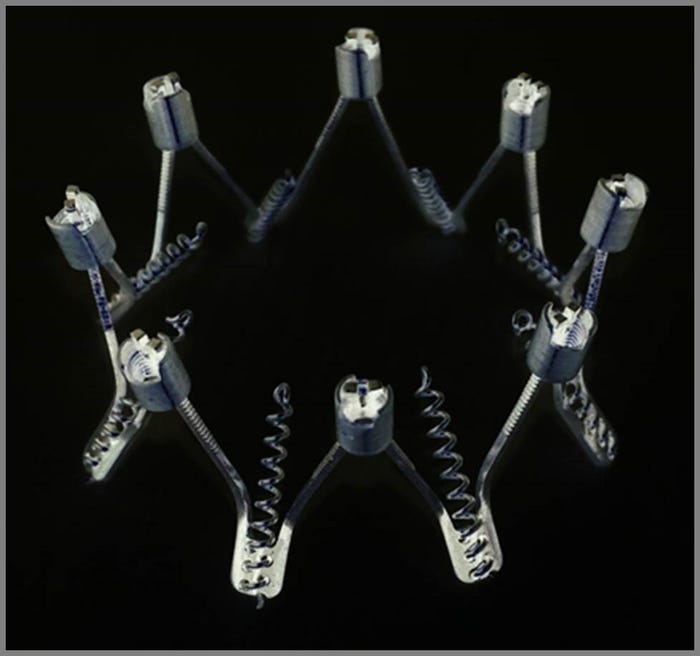
Millipede's 50-millimeter IRIS annuloplasty ring |
Interest in minimally invasive medical procedures continue to grow, and now a wider population of patients suffering from mitral disease may have a new option. Millipede Inc. has expanded its catheter-based mitral implant series to include a 50-millimeter IRIS annuloplasty ring with a transfemoral, transseptal delivery catheter. This device has been implanted successfully in a patient with functional mitral regurgitation (FMR), according to the company.
"The annuloplasty ring is the 'gold-standard' in mitral valve repair," Randy Lashinski, CEO for Millipede, told Qmed. "Millipede has developed the world's first complete annuloplasty ring delivered to the mitral valve via femoral catheter. We are now able to treat a larger population pool with more severe mitral regurgitation."
The FMR patient has often had an "unmet need," Lashinski said. Such patients are "often untreated due to the lack of highly skilled cardiac surgeons and catheter-based tools for the interventional cardiologist. We hope to make this less invasive procedure the first catheter tool the physician reaches for allowing additional therapies in the future if needed for the patient."
When asked whether the use of a transfemoral, transeptal delivery catheter unique for mitral and tricuspid disease is unique, Lashinski said "yes and no. The evolution of delivering devices to the left side of the heart via transeptal access is far safer than surgery, aortic or transapical delivery. Many are now using this access to enter the left side of the heart as structural heart tools are being developed."
Millipede's 50-millimeter IRIS annuloplasty ring is also a repositionable and retrievable device. "Designing in the ability for the physician to reposition and remove the implant, if needed, provides the ability to safely stop the procedure if for any reason at any time before disconnecting from the delivery system," says Lashinski.
"Minimally invasive medical devices are preferred by patients and physicians," he continued. "These include ICDs and stents saving time, cost, and patient morbidity. As technology evolves and advances in medicine, the patient and healthcare sees the benefit in reduced cost and expanded care for patients worldwide. The tools we create today at these early stage companies become commonplace for hospitals around the world."
For more details, visit www.millipedemedical.com.
Daphne Allen is executive editor of Pharmaceutical & Medical Packaging News and a contributor to MD&DI and Qmed. Reach her at [email protected] and on Twitter at @daphneallen
About the Author(s)
You May Also Like



.png?width=300&auto=webp&quality=80&disable=upscale)
