Bard Hopes to Leverage Bos Sci's Heft To Sell Novel Product
C.R. Bard strikes a distribution agreement with Boston Scientific to sell its novel drug-coated balloon catheter to treat patients with peripheral artery disease.
February 10, 2015

Arundhati Parmar
In October, C.R. Bard won FDA approval to commercialize the nation's first drug-coated angiplasty balloon catheter to treat patients with peripheral artery disease.
Less than three months later, Medtronic won its own approval for the IN.PACT drug-coated balloon.
|
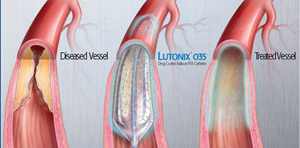
Boston Scientific will distribute Bard's Lutonix DCB |
Now, hoping to cement its first-mover advantage, Bard has announced that it has struck a limited distribution agreement with Boston Scientific to sell its Lutonix DCB. Boston Scientific is a big player in peripheral vascular interventions.
"This agreement is designed to expand the reach of the Lutonix DCB and will afford more patients suffering from peripheral artery disease an opportunity to benefit from this innovative technology and clinically-proven therapy," said Steve Williamson, president of Bard Peripheral Vascular, a Bard subsidiary, in a news release.
The terms of the agreement weren't disclosed.
"Drug-coated balloons are a growing part of the treatment algorithm for peripheral artery disease and provide an alternative in the event a physician chooses not to place a permanent implant or scaffold," said Jeff Mirviss, president, Peripheral Interventions, Boston Scientific, in the news release. "We believe Bard's Lutonix DCB is a great addition to our broad portfolio of vascular products."
In the U.S., Bard's is the first drug-coated balloon used to reopen arteries in the thigh and knee when they become narrowed or blocked because of peripheral artery disease. The drug-eluting balloon is coated externally with the drug paclitaxel that has the potential to help prevent the arteries from being blocked again after the procedure.
This is not the first time that Bard has had a financial transaction with Boston Scientific. In November, 2013, the Marlborough Massachusetts device maker bought Bard's electrophysiology business for $275 million in cash.
You May Also Like

.png?width=300&auto=webp&quality=80&disable=upscale)
