Medical Devices' Silver Bullet
Antimicrobial coatings for preventing infections are becoming indispensable components of implantable medical devices
July 12, 2009
Medical implants have changed the course of healthcare. In the five decades since Swedish scientists invented the first implantable heart pacemaker, medical science has developed implantable devices to treat every conceivable ailment, from hearing impairments and clogged arteries to bad knees and diabetes. Before you know it, a new generation of implants will hit the market.
However, the use of implantable devices is not risk free. The moment a doctor inserts a foreign body in your inner ear or in one of your arteries, infections can develop. This risk is reflected in healthcare statistics: Approximately 80,000 catheter-related bloodstream infections occur each year in U.S. intensive care units, leading to as many as 24,000 deaths. For example, research shows that catheter insertions during continuous ambulatory peritoneal dialysis have resulted in high incidences of peritonitis caused by coagulase-negative staphylococci, Staphylococcus aureus, Pseudomonas aeruginosa, and Candida spp.
To prevent life-threatening conditions, medical device makers are increasingly turning to suppliers that specialize in coatings with antimicrobial properties. At the top of the list of antimicrobial agents is ionic silver, a well-accepted and proven broad-spectrum material. Used to eradicate gram-positive, gram-negative, and antibiotic-resistant bacteria, in addition to yeasts such as Candida albicans, silver is the material of choice for a host of suppliers whose goal is to reduce infections resulting from implantable devices. Complementing the trend toward antimicrobials is the emergence of coatings with antithrombogenic, lubricious, and hydrophilic properties. Taken together, these coatings have improved patient safety and comfort.
Controllings Bugs
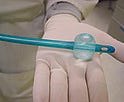
Click to enlarge |
A catheter and balloon for stroke victims from CoAxia Inc. are treated with a Biocoat coating. |
AcryMed Inc. (Beaverton, OR; www.acrymed.com), a provider of antimicrobial surface treatments and wound-care products, has dedicated itself to reducing the incidence of infections associated with implantable devices. "Infection control is what we're all about," remarks AcryMed's president Jack McMaken. Coated on percutaneous catheters and permanent implantable products made from polymers and metals such as cobalt, chrome, titanium, and stainless steel, AcryMed's SilvaGard is used by customers with products on the market and by those seeking regulatory clearance in preparation for future product launches. "Our customers send us their finished products, and we treat them and send them back after making them antiinfective," McMaken says. "Our coating won't allow bacteria to colonize on the device and become a vector for infection."
The company's antimicrobial coating is deposited in the form of metallic silver only. "That's the only thing that remains on the surface of the device," comments McMaken. "After we treat the surface using chemicals in an aqueous fluid, we can deposit the silver. Any surface that contacts the fluid can be treated, whether it's the outside of the device, the inside lumen, any little cracks and crevices. We get down where the fluid goes and deposit elemental silver in tiny particles." This method does not change the nature of the underlying device material.
Unlike AcryMed, many other companies that provide silver-based antimicrobial coatings use some sort of silver salt or silver-complex chemistry, such as silver sulfadiazine, McMaken explains. Generally mixed with a carrier, silver in this form sticks to the surface of the device. Other classes of silver antimicrobials include silver ions encased in zeolite and silver-infused polymers. In the latter case, the polymer material itself undergoes transformation, while a portion of the silver remains on the surface and functions as an antimicrobial agent.
Silver in its metallic form is not antimicrobial, McMaken emphasizes. In the form of Ag0, it is a fully reduced material. But when the outer layer of the metal tarnishes, it forms a layer of silver oxide, which is a very weakly soluble salt. When the silver coating on a medical device dissolves in a fluid, such as the serum in the body or the fluid in body tissues, Ag+ is released. In this ionic form, silver is highly reactive and antimicrobial.
"Our coating is host dependent," McMaken notes. "For short-duration devices, we can put very little on the surface so that it can be used up in three to five days, or we can put a lot on the device so that it will last for months. It's basically a matter of the silver itself--the tiny particles of silver--tarnishing, or oxidizing, on the outside layer." After tissue fluids dissolve the oxide layer, a new layer forms and then dissolves again. The rate at which the silver particles erode depends on how much fluid they are subjected to. The coatings on long-term devices such as bone implants, which encounter small amounts of fluid, may remain in the body for long periods of time.
AcryMed's coatings are used on devices such as a short-duration nylon drug-delivery catheter used for regional anesthesia delivery. Marketed by I-Flow Corp. (Lake Forest, CA; www.iflo.com), the catheter is left in the body for seven to 10 days, requiring that SilvaGard be coated on the inside, the outside, in the lumen, and in the drug-delivery holes. "Back up the vascular access route is a needleless connector for which we provide the surface treatment," McMaken says. Manufactured by Baxter Corp. (Deerfield, IL; www.baxter.com), the V-Link connector is a luer-activated device with a SilvaGard coating that kills at least 99.99% of the six common pathogens known to cause catheter-related bloodstream infections, according to the company.
Disrupting Bacteria
Click to enlarge |
Antimicrobial zones of inhibition produced by silver ions in Covalon's Covacoat process are demonstrated with streptococcal necrotizing fasciitis. |
Biocoat (Horsham, PA; www.biocoat.com) is also no slacker when it comes to silver-based antimicrobial-protected coatings. A provider of coatings for medical products such as catheters and guidewires as well as fertility, respiratory therapy, and ophthalmological devices, the company has partnered with Agion (Wakefield, MA; www.agion-tech.com) to incorporate an ionic form of silver--called silver zeolite--into its coatings.
Silver ions fight microbes by preventing respiration and thereby inhibiting transport functions in the cell wall. They also inhibit cell reproduction and disrupt cell metabolism, states Josh Simon, Biocoat's senior product manager for coating technologies. Zeolite crystals containing silver ions are randomly oriented and distributed throughout the surface of a fiber, polymer, or coating. In conditions that support bacterial growth, positive ions, in ambient moisture, exchange with silver ions at reversible bonding sites on the zeolite. The exchanged silver ions are then available to control microbial growth.
In contrast to other antimicrobial coatings, zeolite is not based on alcohol, chlorine, or ammonium. It works by exchanging silver ions with other positive ions--often sodium--from the moisture in the environment, effecting a release of silver on demand. The multifaceted zeolite crystal carrier provides a 3-D mechanism for releasing silver ions independently of particle orientation in the substrate.
Besides providing silver-based coatings, Biocoat can also attach various polysaccharides to a device surface, such as hyaluronic acid. The purpose of hyaluronic acid, a natural material found extensively within body tissue, is to impart lubricity, biocompatiblity, and antithrombogenicity to the surface of a device. Used in Biocoat's Hydak coatings, hyaluronic acid can also be cross-linked or non-cross-linked on a surface to control durability in applications in which abrasion may occur.
"Our topcoats are made mostly from hyaluronan, which degrades quickly in vivo into nontoxic by-products that are quickly and harmlessly absorbed by the body," Simon comments. "Moreover, hyaluronan is also resistant to protein adsorption and bacterial attachment, which makes it an ideal material for blood-contacting surfaces."
Synergisms
Biocoat's strategy of providing coatings that synergistically combine antimicrobial and antithrombogenic properties reflects an industry trend that is being pursued by other companies as well. For AST Products Inc. (Billerica, MA; www.astp.com), synergy means offering antimicrobial coatings that also render surfaces lubricious, hydrophilic, and antithrombogenic, according to William Lee, the company's director of R&D.
Polymer-based formulations, the company's coatings are suitable for most polymer-based medical materials, including Pebax, nylon, polyurethane, polyethylene, polypropylene, and PEEK. Promising results have also been found on many metals and metal alloys used in the medical device industry, according to Lee. Applied to a range of biomedical devices in the neurovascular, cardiovascular, and urological areas, the coatings are suitable for catheters, guidewires, vascular grafts, intraocular lenses, and other implantables.
"Our antimicrobial coating, RepelaCoat, is a silver-based infection-resistant medical device coating with a patent-pending ion-exchange release mechanism," Lee says. "Unlike most antimicrobial coatings that have only a brief, initial spike of activity, our coating can be customized to have a controlled initial peak release followed by a sustained release of antimicrobial agents in the presence of blood or urine."
Complementing its antimicrobial technology, AST Products' HemoLast coating contains antithrombogenic agents such as heparin, hirudin, or other anticoagulants. Containing both releasable and covalently bonded antithrombogenic agents, the coating provides short- and long-term protection against clot formation on device surfaces, helping to maintain oxygen-exchange efficiency.
"In the real world, there is no universal coating that fits all sizes," Lee says. "The complexity of customizing a specific coating is multiplied by the substrate's composition, geometry, and cleaning and pretreatment methods. It is also affected by the coating methods used, the coating's scalability, and the device's application specifications. As a result, a tailor-made coating for each device is necessary." The importance of surface coatings in the medical device industry to augment medical device performance is becoming better understood, he adds. "Irrespective of the type of application, all medical device coatings are intended to improve the quality of patient care and patient safety."
Shedding Light on Coatings
Click to enlarge |
An all-silicone urological catheter features an expansion of Covacoat on the surface of a fully expanded balloon. |
"In-dwelling medical devices are a crucial lifeline for patients in hospitals," remarks Frank DiCosmo, president and CEO of Covalon Technologies Ltd. (Mississauga, ON, Canada; www.covalon.com). "The devices, often in the form of tubes, provide access to the body for the exit and delivery of fluids, drugs, and even nutrition and air. They also provide a means of removing unwanted substances from the body through urological catheters, kidney dialysis tubes, and wound drains." The problem is, the insertion of such devices provides a conduit for skin bacteria and yeast to gain entry into the body and cause infection.
Engineered using Covalon's Covacoat process, a variety of coatings can be applied to many different medical devices, including central venous lines and Foley, peritoneal dialysis, and hemodialysis catheters. The coatings act as a repository for the controlled efflux of therapeutics at the site of device insertion or application, DiCosmo explains. Once a coating has been applied, therapeutics with different molecular structures can be introduced into the surface matrix. One such therapeutic treatment involves the deposition of silver ions, creating an antimicrobial surface coating that deters the adhesion of microbes to the surface of the device.
Suitable for a variety of polymers, including silicone, polyurethane, and polyvinyl chlorides, Covalon's coatings are applied using a low-energy, surface-modification dip-coating process and ultraviolet light, which induces the growth of the polymers on device surfaces. When activated by ultraviolet light, initiator reagents yield reactive intermediate molecules that remove a hydrogen atom from the polymer surface. The reactive polymer surface then allows monomers in solution to form carbon-carbon or carbon-nitrogen bonds with the polymer device surface via a chain-reaction mechanism. This also causes the monomers in solution to form a covalent polymer coating.
The high reactivity of the initiator reagents makes covalent coating attachment adaptable to many polymer medical devices. The initiator molecules do not become part of the coating but are washed away. Therapeutics can also be introduced in a dip-coating process following the formation of the polymer matrix. "Covalent attachment of the hydrophilic polymer coating molecules is the preferred method of surface modification for polymeric medical devices," DiCosmo remarks. "The coating is readily applied to polymer materials of varying geometries. In addition, the interior and exterior surfaces of tubular devices, as well as films and sheets, can be coated."
Covalon's coating process enables the company to use virtually any type of polymer that is accessible through free-radical polymerization--including polyethylene glycol methacrylates, polyhydroxyethyl methacrylates, polyacrylamides, and a variety of other polyacrylates. Hydrophilic for the most part, these materials endow surfaces with lubricious properties, which can enhance medical device biocompatibility. In addition, being able to control surface chemistry through the choice of an appropriate polymer allows manufacturers to design surfaces that interact with biological components to achieve desired therapeutic effects. "A basic example of this involves coatings that can engage in ion exchange when they come into contact with body fluids," explains DiCosmo. "This property has been exploited to great effect through the use of coatings that can bind antimicrobial silver ions, which are then released locally in a sustained manner at the site of insertion."
Advances in the design of removable and implantable medical devices such as catheters, guidewires, and stents will lead to improvements in diagnostic and therapeutic practice, medical care, and patient outcomes, DiCosmo emphasizes. New devices will have improved features and benefits, and their effectiveness will depend largely on the availability of medical coatings with enhanced properties. Enhancements may include improved antimicrobial activity in response to microbial ingress, as well as improved antithrombogenic and biocompatible properties, and abrasion resistance. For implantable devices such as stents, future improvements in "bioerodability" may enhance their functionality and patient comfort. Other devices may well contain surface coatings that will endow them with "bioactive" characteristics in response to the specific patient and the point of insertion.
"Medical coatings are crucial to the manufacture of advanced medical devices," DiCosmo concludes. "It is expected that there will be an increase in the demand for medical devices, and that will result in accelerated growth for medical coatings." Copyright ©2009 Medical Product Manufacturing News
You May Also Like