Need Info on Surgical Medical Device Materials? Try This.
December 19, 2014
|
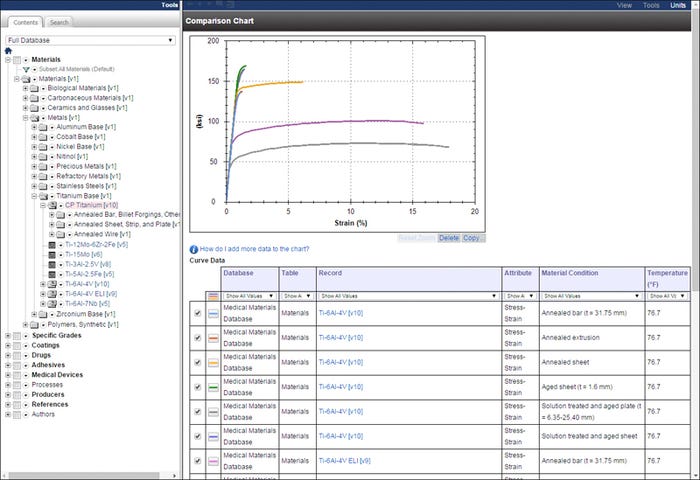
A page from the ASM International/Granta Design database on surgical medical device materials shows stress/strain graphs of titanium alloys. |
ASM International and Granta Design have developed a surgical module in the ASM Medical Materials Database. Focusing on materials used in surgical devices, the module combines engineering material properties and biomedical response data with surgical application information.
The data include descriptions of surgical devices and the materials from which they are constructed, including specific grades and coatings. Coverage also includes materials' mechanical, physical, biological response, and drug compatibility properties. Available through an online web portal or from within a company's in-house materials information system, the module provides:
o Access to materials information, including from predicate devices.
o A means to screen, analyze, select, and source candidate materials and coatings for device applications and compatible drugs.
o The ability to trace all data back to published literature, FDA device approvals information, or manufacturer datasheets and websites.
o Hundreds of material types that are available from more than 1000 providers, together with compatible coatings, drugs, adhesives, and processes.
Device coverage includes more than 33,700 FDA approvals and additional reference material. A range of surgical devices is covered, including those used in diagnostic, prosthetic, surgical, and therapeutic general and plastic surgery applications.
Enabling designers to make informed decisions about material selection, substitution, qualification, and regulation, the surgical module is integrated with existing orthopedic, cardiovascular, and neurological data modules.
Bob Michaels is senior technical editor at UBM Canon.
About the Author(s)
You May Also Like