Gore Scores FDA Nod for More Precise TEVAR Solution
The company said its Gore Active Control System provides controlled, two-stage deployment, with primary deployment to an intermediate diameter and a secondary deployment to full diameter.
May 21, 2019
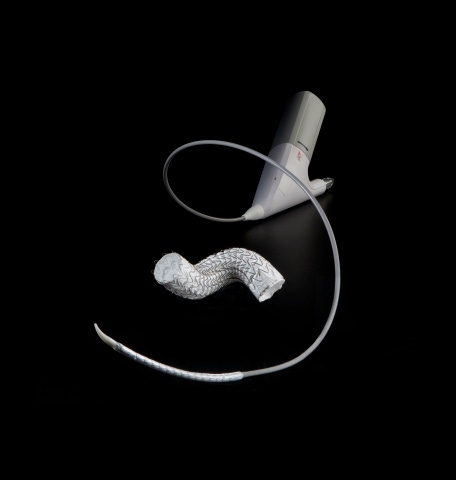
W.L. Gore and Associates has received a nod from FDA for a more precise Thoracic Endovascular Aortic Repair (TEVAR) solution.
The Flagstaff, AZ-based company said the Gore Tag Conformable Thoracic Stent Graft with Active Control System provides new precision and predictable patient outcomes in the endovascular repair of aneurysms, transections, and Type B dissections of the descending thoracic aorta.
A smaller-diameter primary delivery sleeve gives the device and system a lower profile across 10 device sizes.
“Gore has been on the front end and has really defined and established the TEVAR minimally invasive repair of the thoracic aorta going back more than 10 years now,” Eric Zacharias, a vascular business leader at Gore, told MD+DI. “Over that time, we’ve [been able to] broaden the treatment of a range of disease states that need repair of the thoracic aorta.”
Zacharias added, “More recently, the change we made was going after the improvement of the delivery system. The [Gore Tag Conformable Thoracic Stent Graft] is known as a device that treats a wide range of anatomies and fits the anatomy really naturally.”
The Gore Active Control System provides controlled, two-stage deployment, with primary deployment to an intermediate diameter and a secondary deployment to full diameter. This design allows for continuous blood flow throughout the deployment with multiple opportunities to visualize and refine graft placement. It is also designed to allow for angulation of the proximal end of the device for improved seal and apposition.
Gore said that there was no change was made to the stent graft itself TEVAR device, has demonstrated long-term freedom from device-related reintervention 93.1% and low complication rates (zero migrations, fractures, or compressions). Large device oversizing windows were engineered, tested, and proven to accommodate differences in proximal and distal landing zone diameters; a unique 6% to 33% oversizing window allows physicians to choose the optimal radial force to fit patient anatomy and etiology, whether treating a young trauma patient or a fragile dissected aorta.
“TEVAR patients are quite diverse and that’s one of the things that makes them an A-typical population compared to much of the medtech industry,” he said. “Because of that, it really creates some challenges of how we engineer these devices to fit younger patients that need to have these implants in place for decades as well as older patients that need to have it to fit and work effectively with more fragile anatomies.”
The approval comes on the heels of Gore scoring nods from Japan, the U.S., and Europe for its Molding and Occlusion Balloon.
About the Author(s)
You May Also Like


.png?width=300&auto=webp&quality=80&disable=upscale)

