Plastic Breathes Life into Respiration System
November 1, 1999

Plastic Breathes Life into Respiration System
An innovative plastic joint has patients breathing easier.
Made by Dubois Ltd. of Great Britain, a new disposable breathing system features a "Swivel Y" connector made of Carilon polymers, aliphatic polyketones from Shell Chemical Co. (Houston). Replacing a heavy metal and rubber joint, the Swivel Y is "a cost-effective disposable component with the functionality of a heavy reusable Y piece but at the cost of a disposable fixed Y," according to Anthony Fraser, sales and marketing director for Dubois.
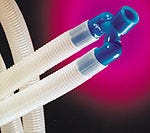 A new disposable breathing system features a Y connector made of Carilon polymers from Shell Chemical Co.
A new disposable breathing system features a Y connector made of Carilon polymers from Shell Chemical Co.
Carilon polymers offer strength and stiffness over a broad temperature range; good toughness, wear, and friction characteristics; and low hydrocarbon permeability. These characteristics allowed Dubois to use the polymers as lips and undercuts for snap-fit assembly without damaging or distorting the finished product. Additionally, the high lubricity of the polymers lets the joint move easily while ensuring a tight fit and minimizing leakage. The polymers are also resistant to chemicals, which reduces stress-cracking problems, and can be sterilized by gamma rays.
For more information about Carilon polymers, visit Shell's Web site at http://www.shellchemicals.com.
Increased Demand Seen for Imaging Equipment, Fetal and Neonatal Monitors
Markets for diagnostic imaging equipment and fetal and neonatal monitors should grow over the next six years, according to two reports issued by Frost & Sullivan, a Mountain View, CA, healthcare marketing and consulting company.
U.S. Fetal and Neonatal Monitoring Equipment Markets analyzes the electronic fetal, neonatal multiparameter, and neonatal noninvasive blood gas monitoring markets. The report forecasts slow growth for sales of the monitors through 2005. It also describes the markets as "moderately to heavily saturated," with companies in the industry focusing on capturing new markets.
The other report, World Diagnostic Imaging Equipment Markets, evaluates the magnetic resonance imaging (MRI), ultrasound, computed tomography, x-ray, and nuclear medical imaging equipment segments of the diagnostic imaging marketplace. Overall, Frost & Sullivan expects industry revenues to exceed $14.6 billion by 2004. Sales of MRI equipment in particular are expected to increase due to improvements in technology. According to the report, the MRI industry will experience an 11% annual revenue increase through 2005, to more than $3.6 billion.
For more information on the reports, visit Frost & Sullivan's Web site at http://www.frost.com.
Medtronic, Xomed to Merge
Medtronic Inc. (Minneapolis) and Xomed Surgical Products Inc. (Jacksonville, FL) have signed a merger agreement under which Medtronic will acquire all shares of Xomed, a leading provider of surgical products used by ear, nose, and throat surgeons. The transaction is valued at approximately $800 million, with Xomed shareholders to receive $60 in Medtronic stock for each share of Xomed. The companies expect the transaction to be completed by the end of the year.
"Xomed's market leadership, strong clinical relationships, highly regarded management, and full range of products offer Medtronic a new platform for continuing growth beyond our current franchises," said William W. George, chairman and chief executive officer of Medtronic, which makes a wide variety of medical devices.
New Technology Aims to Slash Needle Sticks
BTG plc (London) has made available for licensing a new technology that could reduce the approximately one million needle-stick injuries reported in the United States each year. Developed by two experts in the field of hydrophilic polymers, the technology uses such polymers to cover the point of a needle and thereby render it harmless.
Once a needle is used, the polymers absorb moisture from the injected solution or surrounding fluid. This causes the polymers to expand and thereby blunt and block the needle, preventing its reuse and virtually eliminating the possibility of a needle-stick injury, according to BTG.
Since no special needle-retraction mechanism is needed, the technology can be used with a conventional syringe. This makes it easy and inexpensive to use and potentially adaptable for almost any needle, BTG claims.
For more information, visit BTG's Web site at http://www.btgplc.com.
Company to Develop Tools for Making 'Mesoscale' Electronic Devices
As part of a four-year, $7.5 million project, the Defense Advanced Research Projects Agency (DARPA) has awarded Potomac Photonics Inc. (Lanham, MD) a contract to develop and commercialize a laser-based tool for the fabrication of next-generation passive electronic devices. The award also grants Potomac full commercial rights to the tool.
The award is part of DARPA's Mesoscopic Integrated Conformal Electronics (MICE) program, the goal of which is development of a commercially available machine capable of producing "mesoscale" electronic devices in a conformal manner on virtually any substrate. Larger than silicon chips but smaller than printed circuit boards, mesoscale electronic circuits promise to allow further miniaturization of medical and other devices.
 Approximately 40 µm wide, this conductive gold medal line was deposited using a laser technique being developed by Potomac Photonics Inc.
Approximately 40 µm wide, this conductive gold medal line was deposited using a laser technique being developed by Potomac Photonics Inc.
Potomac's MICE tool showcases the company's experience in laser microfabrication technology, as well as a proprietary deposition process developed at the Naval Research Laboratory in Washington, DC. This combination has already resulted in the fabrication of conductors, resistors, capacitors, and inductors.
For more information, call Potomac Photonics at 301/459-3031.
In BriefHill-Rom (Batesville, IN), a manufacturer of patient care systems, has acquired Amatech Corp. (Acton, MA), a manufacturer and distributor of surgical table accessories and patient positioning devices...Tekni-Films, Tri-Seal, Plastron/Natvar, and Colorite Polymers have combined to form Tekni-Plex Health Care Group (Somerville, NJ). The new company is a diversified manufacturer of packaging products and materials for the healthcare industry...The Health Industry Manufacturers Association (Washington, DC) announced its approval of the decision of the National Toxicological Program to reexamine the potential reproductive and developmental effects of seven phthalates. One phthalate, DEHP, is used in a variety of medical devices, including blood bags, intravenous tubing, and respiratory equipment...Colorado MEDtech Inc. (Boulder, CO) has acquired the assets of Creos Technologies LLC (Englewood, CO), a provider of x-ray generator subsystems for the medical imaging industry, for approximately $2 million...Tyco International Ltd. (Hamilton, Bermuda), a diversified manufacturing and service company, has agreed to acquire General Surgical Innovations Inc. (Cupertino, CA), a medical manufacturer and distributor of balloon dissection devices. The value of the transaction is approximately $100 million...GE Marquette Medical Systems (Milwaukee), a provider of medical diagnostic equipment, has signed a definitive agreement to acquire Prucka Engineering Inc. (Houston), a company that specializes in electrophysiologic research and development, for an undisclosed sum...Merit Medical Systems Inc. (South Jordan, UT) has acquired the catheter unit of Mallinckrodt Inc. (St. Louis). The catheter plant, located in Angelton, TX, manufactures diagnostic and interventional catheters for cardiology and radiology procedures...Norton Performance Plastics Corp. (Wayne, NJ), a manufacturer of specialty polymer products, has acquired Sanitary Couplers (Springboro, OH), a manufacturer of reusable couplers for hoses. The company will be a part of Norton's PPL Fluid Systems business...Guidant Corp. (Indianapolis), a manufacturer of products for the treatment of cardiovascular and vascular disease, will acquire CardioThoracic Systems Inc. (Cupertino, CA), a cardiac surgery instrument and systems company. The transaction will be a tax-free exchange valued at approximately $313 million...BD (Franklin Lakes, NJ), formerly Becton Dickinson and Co., has acquired Saf-T-Med Inc. (Chicago), the maker of a safety-engineered syringe and other needle-based medical devices...Biomet Inc. (Warsaw, IN), an orthopedic device manufacturer, has agreed to merge with dental implant manufacturer Implant Innovations Inc. (Palm Beach Gardens, FL). The transaction, to be completed by late November, is valued at approximately $175 million...MedSearch Technologies Inc. (New York City) has completed its acquisition of M&W Medical Supply LLC, a research and development company...Phillips Petroleum Co. (Bartlesville, OK) and Solvay Polymers Inc. (Houston) have agreed in principle to build and operate a high-density polyethylene manufacturing facility in the United States...St. Jude Medical Inc. (St. Paul, MN) has signed a definitive agreement to acquire Vascular Science Inc., a Minnesota-based developer of suture-free devices for coronary artery bypass graft anastomosis, for $80 million in cash and $20 million in milestone payments. |
Copyright ©1999 Medical Product Manufacturing News
You May Also Like
.png?width=300&auto=webp&quality=80&disable=upscale)

