Opioid Wearable Opens a Large Market Opportunity for Masimo
FDA has granted marketing authorization for the Masimo Opioid Halo, a wearable system designed to detect opioid-induced respiratory depression.
April 5, 2023
In 2016, Yvonne Gardner, a Vernal, UT resident, experienced something no parent should ever have to endure. Her son, Parker Stewart, died after taking prescription opioids to manage his pain after a tonsillectomy. Now, a new wearable opioid overdose prevention device could help other parents from losing a child to opioid-induced respiratory depression, Gardner says.
FDA granted a de novo authorization for the Masimo Opioid Halo, the Irvine, CA-based company recently reported. With this regulatory milestone, Masimo also becomes the first winner of an FDA Opioid Innovation Challenge to have an authorized solution designed to help address the U.S. opioid crisis.
The authorization also comes on the heels of FDA approval to sell Narcon without a prescription. Narcon is the leading version of the opioid overdose-reversing drug naloxone.
The de novo authorizes Opioid Halo to be made available over the counter without a prescription, for use on adults and children age 15 and up, and an Rx version for use by prescription from a healthcare provider. Opioid Halo advances the forefront of continuous monitoring through its Opioid Halo engine, an advanced pattern recognition algorithm which helps detect and quantify the risk of severe opioid-induced respiratory depression, Masimo noted. Combined with its distributed architecture, Opioid Halo is designed to help manage and send escalating alarms to family members, friends, and caregivers, notifying them that help may be needed due to an opioid overdose – including triggering an automatic wellness call, which may lead to emergency medical services being dispatched.
"We are very excited to be able to offer this solution to our fellow Americans and the community heroes who are helping to battle the opioid crisis – a crisis so devastating in its impact on the young that it has lowered overall life expectancy in the U.S.," said Joe Kiani, Masimo's founder and CEO. "Now, with Opioid Halo, we hope to help make a big difference by providing a much needed tool that can help millions, whether they are taking prescribed opioids or struggling with illicit opioid use."
Masimo was chosen by FDA in 2018 to develop a device that could help prevent opioid overdose, as part of the agency's Opioid Innovation Challenge.
"Between then and now, the COVID-19 pandemic accelerated our development and refinement of the Masimo SafetyNet family of remote patient management and telehealth solutions, helping save many COVID patients’ lives," Kiani said. "We thank the FDA for taking on the opioid epidemic and granting this de novo – a clearance that is a huge step forward in preventing overdose deaths and helping to end the opioid crisis.”
According to a report by Marie Thibault, a medtech analyst at BTIG, Masimo had previously noted a potential U.S. market as large as $4.5 billion for SafetyNet Opioid based on the roughly 25 million patients taking prescription opioids for post-operative or chronic pain.
"It is unclear if the planned go-to-market strategy has remained the same, but the company expected to focus on surgeons and pain specialists, both high-volume prescribers of opioids, as the appropriate sales target," Thibault wrote. "We expect that [Masimo] will seek reimbursement, since management had previously noted that existing reimbursement codes may cover limited use cases but that they would also plan to apply for new codes. Additionally, [Masimo] had launched the SafetyNet Opioid System in a few European countries, but management has previously described the early response as underwhelming."
Opioid overdose is the leading cause of accidental death in the United States, responsible for more than 80,000 of the roughly 100,000 drug-related deaths in 2021, from both illicit opioids, such as fentanyl and heroin, and prescription opioids. Masimo noted that more than 143 million opioid prescriptions were written in 2020 to help people manage pain. But these pills come with a dangerous risk of opioid-induced respiratory depression – slowed or stopped breathing – which is the leading cause of death from opioid overdoses. Anyone taking opioids, prescription or illicit, is at risk of experiencing an accidental overdose, the signs of which are unpredictable and can be difficult to detect, Masimo said.
How does the Opioid Halo system work?
Masimo Opioid Halo is designed to help family and friends identify the symptoms of an opioid overdose by detecting physiological markers present during opioid-induced respiratory depression and ideally, helping them know when it’s time to intervene – for example, by administering a potentially life-saving dose of naloxone. Opioid Halo can be used at home or in the hospital or another care setting, by patients prescribed opioids after surgery or managing a chronic or prolonged condition, as well as people suffering from opioid use disorder.
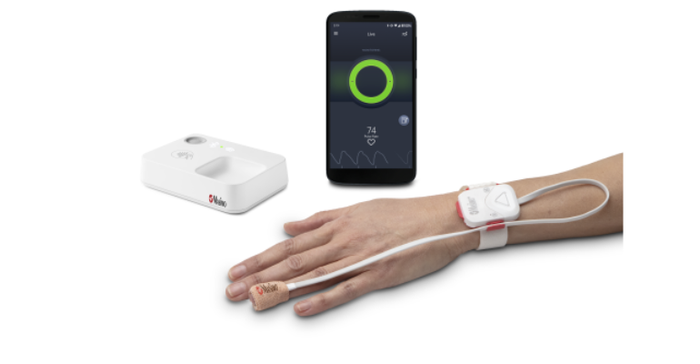
The Opioid Halo system consists of four components: a tetherless, adhesive fingertip sensor; a reusable Masimo SET pulse oximeter and Bluetooth chip; a Bluetooth-to-Wi-Fi Masimo Home Medical Hub; and a smartphone app. The fingertip sensor provides real-time monitoring for opioid-induced respiratory depression, enabled by the Opioid Halo pattern recognition algorithm and Masimo SET (signal extraction technology) – even during movement, when hands are cold, and on all skin pigmentations. Data from the sensor and chip are wirelessly relayed to the Masimo Home Medical Hub and the smartphone app, which continuously analyzes the user’s physiological data for trends and patterns associated with the physiology of an opioid-induced respiratory depression event to quantify the risk of an opioid overdose. As the level of risk rises, the app and hub provide alerts. Upon early onset, an audible and visual alarm, designed to trigger early intervention opportunities for the user to self-recover or get help, is provided. If the Opioid Halo risk score continues to worsen, in addition to the repeated alarms, automatic texts are sent to designated friends and family members, letting them know it may be time to intervene, for example by administering naloxone or taking other action. Finally, if the severity of the risk level progresses even further, there is an optional setting that can be activated during setup that enables a service center to place an automatic wellness call to the user, the outcome of which may lead to emergency medical services being dispatched.
"Opioid Halo is another breakthrough innovation that can help save lives by adding to the critical support tools for people who use drugs," said Ryan Hampton, an advocate, author, and person in recovery.
About the Author(s)
You May Also Like



