Bethany Corbin Women's Health Thought Leader
Manufacturing
FemInnovation CEO to Keynote IME SouthFemInnovation CEO to Keynote IME South
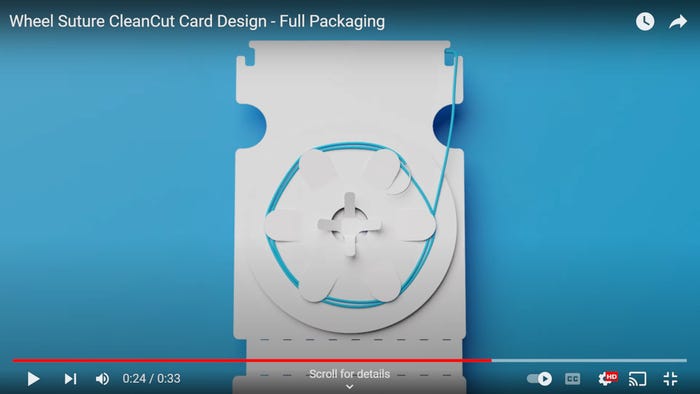
Bethany Corbin, a recognized thought leader in women's health, is set to keynote IME South. The six-in-one advanced design and manufacturing expo returns June 4-6 to the Charlotte Convention Center.
Sign up for the QMED & MD+DI Daily newsletter.








.png?width=300&auto=webp&quality=80&disable=upscale)




.png?width=300&auto=webp&quality=80&disable=upscale)







.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)








.png?width=300&auto=webp&quality=80&disable=upscale)