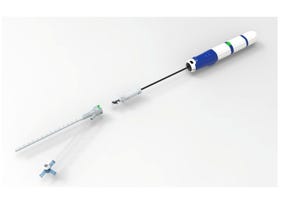
Mesh Suture noted that the aim of DURAMESH is to mitigate the intractable problem of surgical failure due to suture pull-through.

Mesh Suture, d.b.a. MSi, today announced the FDA 510(k) clearance for DURAMESH, a non-absorbable polypropylene mesh suture used for the surgical closure of soft tissues including muscles, fascia, tendons, and ligaments. The suture, according to the company, combines the desirable principle of implant incorporation in a mesh repair with the placement precision of a suture for soft tissue repairs.
“DURAMESH offers the perfect combination of strength and simplicity in a surgical repair,” said Gregory Dumanian, MD, CMO at MSi. “It combines the handling characteristics of traditional suture with a mesh polyfilament design.”
The aim of DURAMESH is to mitigate the intractable problem of surgical failure due to pull-through. Additionally, the sharp leading edge of a traditional suture can slice otherwise intact tissue, causing potential dehiscence, hernia formation, and poor tendon function. In comparison, DURAMESH uses an open-walled hollow design that allows tissue ingrowth for implant incorporation without capsule formation and flattens at the suture-tissue interface to resist pull-through.
“We are excited to bring this innovative technology to our surgeon colleagues and to their patients who need it most,” Dumanian said. “By designing DURAMESH to address the surgical complication of suture pull-through, we expect to see sizeable improvements in patient outcomes.”
Currently, DURAMESH is already in clinical use in the EU and UK, having received CE Mark designation in the second quarter of 2021.
About the Author(s)
You May Also Like