A new study offers more evidence that single-use bronchoscopes could be a better alternative to reusable devices.
July 12, 2018
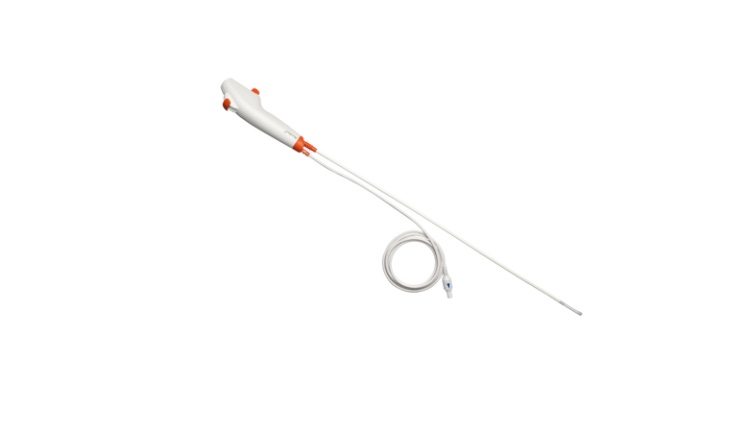
A recent study, funded by 3M Company and supported by Healthmark Industries, found that most of the reusable bronchoscopes it examined, even when cleaned and disinfected using current reprocessing guidelines, remained contaminated or damaged.
“The results are worrisome, as patients undergoing bronchoscopy are commonly at high risk for infection due to transplant status, critical illness, or immune-suppression due to malignancy or chronic disease,” states the study, “Effectiveness of Reprocessing for Flexible Bronchoscopes and Endobronchial Ultrasound Bronchoscopes."
The 2017 study was conducted at three large hospitals in the United States, including two transplant centers. The researchers examined 24 clinically used flexible bronchoscopes after they had been cleaned and disinfected adhering to each facility’s institutional practices.
Contamination was detected using microbial cultures and tests for protein, hemoglobin, and adenosine triphosphate. After manual cleaning, the researchers found all the bronchoscopes were still contaminated. The study says microbial growth, including that of mold, Stenotrophomonas maltophilia, and Escherichia coli/Shigella species, was found in 14 fully reprocessed bronchoscopes (58% of the total).
Visible irregularities were also seen in all the bronchoscopes, including retained fluid; brown, red, or oily residue; scratches; damaged insertion tubes and distal ends; and filamentous debris in channels.
The study concluded by saying that damaged and contaminated bronchoscopes were in use at all three sites, noting that inadequate reprocessing practices may have contributed to this. Two of the three study sites had substandard reprocessing standards.
However, even when reprocessing guidelines were followed, high-level disinfection was “not effective,” the researchers said. They recommended a shift toward the use of sterilized bronchoscopes, as well as updating quality management programs and reprocessing guidelines.
These results seem to provide confirmation of various other studies that have been done with endoscopes, not specifically bronchoscopes. “This study was specifically done for bronchoscopes because that data was lacking,” says Lars Marcher, CEO and President Ambu A/S, in an interview with MD+DI.
Contamination of reusable endoscopes has been known for some time now. “Endoscope Reprocessing Failures Continue to Expose Patients to Infection Risk” was number two on ECRI Institute’s list of Top 10 health technology hazards for 2018. And it has been on the organization’s top 10 list for the past eight years.
Shifting to single-use instruments will likely be cost effective as well. According to a 2017 study, “Early Assessment of the Likely Cost Effectiveness of Single-Use Flexible Video Bronchoscopes,” the estimated cost of using reusable bronchoscopes is $424, with a 0.7% risk of infection. Single-use technology costs an average of $305, with 0% risk of infection.

“Flexible endoscopes raise specific problems because they are costly to purchase, reprocess, and repair; they are not always available when you need them; and their use risks exposing patients to infections,” says Ambu’s Marcher. “We believe that the challenges in flexible endoscopy should be addressed with single-use devices, and for more than a decade we have been harnessing the technology.”
Ambu offered the first disposable airway endoscope approved for sale in the United States for airway management in April 2010. A second generation in 2011 was optimized for intubating. The third-generation disposable airway endoscope targeted bedside bronchoscopy. It has a suction channel and three sizes—large, regular, and slim. And the fourth generation offers an upgraded image quality and a more robust design. (See packaged device at right.)
The company’s bronchoscopes are used for applications such as intubation, bronchoalvelolar lavage and bronchial wash, airway inspection, tube checks, protected brush sampling, percutaneous dilatational tracheostomy, and for training in bronchoscopy skills.
About the Author(s)
You May Also Like




