April 19, 2010
|
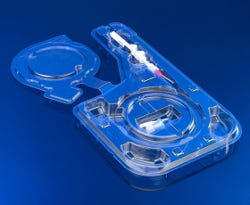
Prent's one-piece universal package design helps customers reduce tooling, per-package, and assembly costs. |
Prent Corp. (Janesville, WI) has won an AmeriStar Award for medical device package design from the Institute of Packaging Professionals (Naperville, IL). The sole winner in the medical device category, Prent's new package design was developed to help a customer reduce tooling, per-package, and assembly costs.
The company's universal package can accommodate 35 stock-keeping units into a single package. "As we continue making refinements in our medical device line, Prent's universal package will easily accommodate both old and new devices, including those with different handles, triggers, and product configurations," remarks the device company's product development engineer overseeing the packaging project.
"Prent's package design is also helping us reduce costs three ways," she explains. "First, tooling costs have been slashed because the package is designed to be thermoformed using just one tool die--rather than separate tools for a tray and retainer. Second, our cost per package has been cut by more than 25% because there's just one thermoformed clamshell tray--rather than a tray and retainer. Third, less assembly time is now required, since the one-piece thermoformed clamshell can be loaded faster."
In addition, the design and location of the fold-over hinge is strategically placed and made as small as possible to eliminate pouch abrasion.
The one piece design, coupled with four undercut features inside the circular cavity, has improved the ease and control of loading and unloading delicate spiral medical devices, according to the company. The undercuts also reduce product damage and prevent the medical device from popping out of the package or out of the operating room's sterile field--a common occurrence, the product development engineer notes.
About the Author(s)
You May Also Like