Intersect ENT has introduced a new kit combining the Propel Mini Sinus Implant and the new Straight Delivery System, based on user feedback from the field.
March 10, 2021
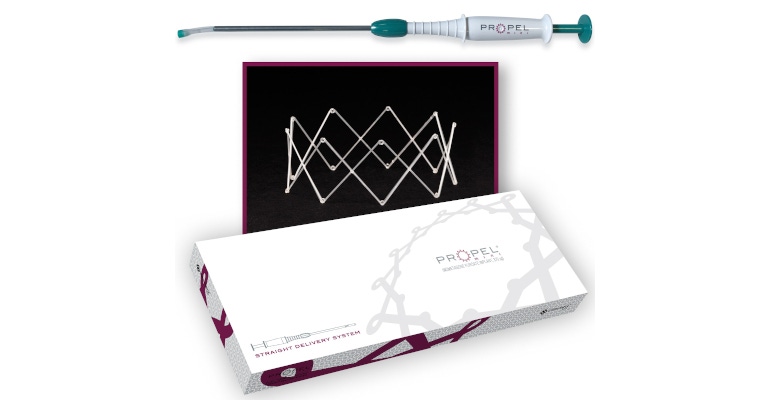
In February, Intersect ENT Inc. debuted a new kit in the United States that combines the company’s new Straight Delivery System (SDS) with its Propel Mini Sinus Implant. The launch evolved from what Intersect ENT president and CEO Tom West calls “customer-led innovation” as well as company-wide teamwork.
The SDS itself received PMA in July 2019, and it had been developed after surgeons requested an additional delivery system for the bioabsorbable polymer sinus implant. The original delivery system is curved and suited for most anatomies of the ethmoid sinus and frontal sinus ostia, but some surgeons had asked for a straight delivery system for the ethmoid sinus.
Intersect had packaged the Propel Mini implant with the original curved delivery system beginning in 2012. After surgeons expressed interest in having a delivery system optimized for the placement of Propel Mini in the ethmoid cavity, the company developed and launched the Propel Mini SDS combination package. The full Propel product line now includes four different kits, indicated for the frontal, ethmoid, and maxillary sinuses: the Propel Mini with the curved delivery system, the Propel Mini with SDS, the Propel Contour with the Flextip delivery system, and the Propel with a straight delivery system.
“Intersect ENT is pleased to provide our physicians with a complete package of Propel Mini and the Straight Delivery System,” West said in a statement. “We are dedicated to responding to our customers’ feedback and providing them with the best combination of products to advance care for chronic sinusitis patients in various centers of care.”
The company shared the following feedback from Roheen Raithatha, MD, a practicing rhinologist at ENT and Allergy Associates and faculty member at Mt Sinai Hospital in New York: “The combination of the Propel Mini and the Straight Delivery System in one package is reflective of Intersect ENT’s ongoing commitment to supporting the ENT community. I look forward to utilizing this newly bundled implant and delivery system during my FESS procedures.”
The kits present the implant fully expanded and provide users with a crimping device to load the implant onto the delivery system. “When the delivery system goes into the nasal cavity, the system places the stent, which then expands to fit, providing dual benefit,” West told MD+DI. “The implant delivers drug onto the nasal tissue, and the radial expansion provides radial flow of air to promote healing.”
The combo packages are each color coded to denote size and model. he explained. “We do that so the surgeon can recognize which one to select. We’ve heard that physicians have come to know the products by their color coding. Such identification is a big benefit.”
West said that as the company looks at further line extensions, it will continue to be informed by the needs of customers. “Our extensions are customer led, not technology led. Not, ‘Here’s our technology—how can you use it?’ It’s not a unique approach, but it is definitely the best practice.

“Feedback comes directly from the field and field representatives gowned up in the surgical suite with surgeons. They learn by watching how clinicians use products during procedures,” West continued. The company also gathers more statistical feedback through surveys. Such work “really helps us understand the job that is to be done and how technology can help,” he said.
The Intersect ENT team “brings in engineering early on during the product development process, including packaging engineers,” West said. “We have to be able to scale and manufacture reliably, and all that requires integrating quality and packaging early. Engineers from operations and regulatory professionals, too. We engage the full organization, and the more integrated we are, the stronger the innovation is. The entire organization plays a role in innovation.”
In related news, the Centers for Medicare and Medicaid Services (CMS) announced in January that it had created separate codes for the Propel line (S1091) as well as for its SINUVA bioabsorbable implant line (J7402). (A single code had previously covered both products despite their differences.) CMS has also attached an average selling price to J7402, “mometasone furoate sinus implant, (sinuva), 10 micrograms,” the company shared in a news release.
About the Author(s)
You May Also Like




