R&D DIGEST
October 1, 2006
|
PDT drugs fluoresce red in UV light as a magnet (left) attracts the nanocarriers. |
Work has been done on developing drug-filled nanocarriers via a magnetic field, but a more concentrated approach to targeting tumors, which could reduce side effects, may be needed. Research at the State University of New York (SUNY) in Buffalo shows promise.
A team at SUNY Buffalo has devised a delivery system that could lower the amount of photodynamic therapy (PDT) drugs that collect in normal tissue.
An external magnetic field is used to steer the accumulation of nanocarriers into tumors. To make the delivery system react to the field, iron oxide nanoparticles are encapsulated in the nanocarriers. Initial studies have shown that the magnetic field causes a localized buildup of the PDT drug in the tumor site. “This system is intended for PDT of cancer but can be, in principle, applied to the delivery of chemotherapy drugs or any other kind of drug for cancer or other diseases,” says Paras Prasad, PhD, professor of chemistry, physics, electrical engineering, and medicine at SUNY Buffalo.
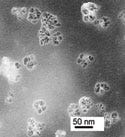
The drug-delivery system is made of a stable aqueous suspension of particles with diameters less than 50 nm, iron oxide–based magnetic nanoparticles, photosensitizers, and polymer micelles. By freeze-drying them into powder form, the particles can also be dispersed at a later time by adding aqueous buffers.
|
A transmission electron microscope (TEM) depicts the carriers. |
“The advantages involve stable aqueous suspension of photosensitizers; ease of storage, transport, and reconstitution into parenterally injectable formulation; as well as feasibility of specific targeting to tumors using external guiding with a magnetic field,” says Prasad, who is also the executive director of the university's Institute for Lasers, Photonics, and Biophotonics. “This magnetic targeting is expected to significantly enhance the localization of the photosensitizer drugs into tumor sites in vivo as compared with nontargeted formulation.”
The system can also be generalized to target chemotherapeutic drugs. Another option is to equip the system with small tumor-targeting molecules such as folic acid and antibody fragments. “Magnetic targeting can guide the nanoparticles until the intravascular spaces near the target tumor,” says Prasad. Therefore, the combination of magnetic guidance and small-molecule targeting can form a synergy to destroy tumors more effectively.
Extensive in vivo testing of the nanocarrier system still needs to be performed. “Because the nanocarriers proved to be significantly stable to retain the PDTs, they can also be used to deliver a wide range of therapies to tumors or other disease sites in the body, without significant loss in the circulatory system or in normal tissues,” says Prasad. In order to extend the approach to other treatments, the researchers are planning to study the nanocarriers' stability and how changing its formulation controls the kinetics of the drug release.
The researchers don't currently envision using the nanocarrier system with gene delivery.
Research was funded by the New York State Center of Excellence in Bioinformatics and Life Sciences, and the university's Interdisciplinary Research and Creative Activities Fund. More details about the technology can be found in the August 2006 issue of Molecular Pharmaceutics.
Copyright ©2006 Medical Device & Diagnostic Industry
About the Author(s)
You May Also Like