Benefits to the method include fast product development and time to market.
March 14, 2012

Reducing healthcare costs is an often-heard battle cry in today’s global economy and is a strong motivator for the move from traditional surgical techniques to less-invasive vascular interventions. For such interventions, medical devices and diagnostic tools are getting smaller, reaching the physical dimensions of an artery or vein, a blood cell, a tumor break-off, or even a DNA fragment. To produce such small and microsized devices, engineers are turning to laser micromachining to reduce development and device costs and get products to market fast.
|
For the basics about laser micromachining, check out “Fundamental Principles of Laser Micromachining of Polymers." |
For example, in vitro diagnostics (IVD) are tests performed in laboratories or at the point of care to allow doctors to monitor the effectiveness of a treatment and to ascertain the correct therapy. IVD instruments, which collect and analyze biological samples (tissue, fluids) for the diagnosis of pathologies, require miniature sensors to detect a condition. While the actual method of sensing varies from disease to disease (diabetes, molecular diagnostics, clinical flow cytometry, etc.), common engineering problems for such sensors include:
Extremely low signal strengths (e.g., electrical current, fluorescence, etc.).
Volume/area regulation fluids samples (such as blood), requiring microsized apertures with micron tolerances.
Miniaturized electrical circuits.
In the case of diabetes, according to the CDC’s National Diabetes Fact Sheet of 2011, diabetes affects 8.3% of the U.S. population, or a staggering 25 million people. Of those diagnosed with diabetes, both type 1 and type 2, 26% take insulin or a combination of insulin and oral medication.
Short-term and long-term implantable sensors, both minimally-invasive and noninvasive, measure glucose levels directly or indirectly by a variety of methods. These polymer-based sensors, in sheet, wire, cable, or catheter format, have a common requirement to regulate the flow of blood, perspiration, and optical fluorescence with apertures or openings with tolerances as small as a few microns. Since the absolute signal produced is proportional to the actual sensor area, laser micromachining fabrication technology is a suitable choice due to its high precision and repeatability.
As a rule of thumb, the smallest feature size achieved by laser micromachining is on the order of couple of the laser wavelengths. As an example, an excimer laser operating at 193 nm, can create a theoretical spot size of approximately 0.4 µm. This laser is also used to fabricate microcomputer chips by excimer lithography. However, the material interaction of the laser beam with the actual polymer may broaden the smallest feature beyond 1 µm.
Since lasers are noncontact tools that do not wear like mechanical counterparts such as drills or dies, machining produced by lasers is very repeatable. For example, if a sensor requires the sensing area to be 50 µm in diameter and the diameter varies by 10 µm (20%), when using drills or dies (for example), the total area changes by 44% (square) which may be unacceptable to the sensor designer. Laser microfabrication addresses this issue by careful optical beam design performed by optical engineers and physicists who balance the theoretical beam propagation models against real-world part tolerances.
Required features in sensors can be laser drilled, laser stripped, or laser cut by a variety of laser sources and techniques. In some cases, the sensor volume is important, because it requires a method to etch down to certain depth and stop. Some lasers can etch as small as 0.1 µm at a time, providing excellent depth control. At the same time, such a “fine scalpel” can also be automated through a variety of part handlers such as pick-and-place, reel-to-reel part handler autoloaders so that manufacturing can be scaled up cost effectively.
Diagnostics
There are multiple areas within the IVD industry in which laser micromachining may be used to fabricate consumable sensors, including cytology, flow cytometry, and molecular diagnostics. Since laser micromachining can drill a hole as small as 1 µm in diameter, lasers are ideal tools for drilling precise holes in polymer-based microfluidic chips to isolate individual cells such as white blood cells or cancer cells. Similar to the cell phone industry, lasers are also used to drill small interconnects and upstream filters.
Cytology involves the study of cell structures, cell composition, and cell interaction. In its simplest form, cell counters are used in life science research to count red and white blood cells to assess a patient’s overall health. Also, the concentration of bacteria, viruses, and other pathogens in blood can help assess the state of an infectious disease or the success of an immune system’s reaction.
Low cost, portable automated cell counters based upon the Coulter counter principle have been developed where a critical opening, for the passing of a single cell through a pair of electrodes, is machined by a laser. The passage of the cell instantaneously increases resistance between electrodes and this gives an indication of the presence of the cell and thus, can count the cells (as well as determine cell volume).
Flow cytometers are sophisticated cell counters that can also analyze internal structures, protein levels, and biochemistry of the cell. Like most instruments, there is a trend to replace table top bulky flow cytometers with handheld portable devices where a small semiconductor laser interrogates the cell passing through a laser-drilled opening. Specifically, the semiconductor laser is pointed at a cell stream tagged with a fluorescent reagent and the reflected, transmitted light, and fluorescence signals are detected. In these applications, lasers are used as both a component and a method of fabrication.
Molecular diagnostics identifies genetic or protein activity patterns to determine the presence of infectious and genetic diseases as well as to identify the types of antigens attacking the host. The measurement of genetic content (DNA, RNA, proteins, and metabolites), including DNA sequencing, may help physicians understand the probability of a patient developing the disease or accepting a specific treatment. The discovery of new genes and biomarkers accelerates the development of new clinical tests. Lasers can be used for fabricating microfluidic blind channels, micropore filter arrays, or apertures for individual cell isolation.
Cancer diagnostics tools can look at the signal transduction at the cellular level to identify specific markers that help the decision path for physicians. Methods for the collection of live tumors are a challenge that may involve laser drilling due to its precision to create openings at the cellular level. Collection of blood to analyze circulating tumor cells is another approach where the detection of fragments from the tumor offers both prognostic and diagnostic applications. Lasers can be used for fabrication of electrical via connections or filters in the disposable chips.
The trend towards low-cost, polymer-based disposables for point-of-care diagnostic tools requires devices for screening and collection of cells as well as miniature electrical circuits to detect and amplify signals. Highly precise laser technology is an attractive manufacturing method to meet the feature sizes and tolerances at the cellular level.
Embolic Protection Devices
|
Polymer-based embolic protection filters have thousands of laser-drilled perfusion openings that enable blood flow while also capturing emboli. |
Embolic protection devices are used in conjunction with vascular intervention procedures to capture dislodged emboli. These embolic events may occur during a stenting procedure such as saphenous vein graft or carotid artery stent procedures and vascular interventions such as a heart valve replacement or the prevention of pulmonary embolism via a peripheral vascular intervention. These polymer-based embolic protection filters, fabricated as 2-D filter screens or 3-D “socks,” contain thousands of laser-drilled perfusion openings that are large enough to allow blood flow through the filter but small enough to capture emboli that if left unattended, could cause an embolic stroke or pulmonary embolism.
There are a number of design considerations when laser drilling thousands of openings of approximately 100 µm in diameter in thin polymer films. Some embolic filters are fabricated from balloons where the 3-D geometry poses challenges since the balloon is nonplanar, especially at the transition from the neck to the body. In this cone region, as steep as 45°, advanced autofocus and balloon-shape mapping techniques are required since the laser beam gets out of focus as the height of balloon surface changes. In filters made of thin and flimsy polymer films, as more openings are laser drilled, the polymer film shifts and deforms as the device loses mechanical integrity. Techniques such as cryogenic processing are deployed when the polymer material is frozen, transforming its material behavior from a flexible polymer to a stiff solid, permitting the opening/hole population to increase without any material shift that could affect subsequent positioning and size tolerances.
Embolic protection devices do not always have to cross the arterial lesion. Balloon aspiration devices, placed proximal to the lesion, act like vacuum cleaners and remove the emboli. These devices may momentarily stop blood flow to aspirate the emboli. There are no micropores here, but like in many interventional devices, lasers are used to strip a polymer layer of a guide catheter to improve flexibility, which is especially useful for passage through tortuous vasculature.
The fabrication of an array of microopenings in polymer films is also applicable to drug delivery. Pulmonary oral drug delivery devices or transdermal patches can benefit from arrays of microopenings because the laser-drilled polymer films can be more cost effective than using holed inserts or EDM-drilled metal foils. Laser-drilled membranes can offer benefits over weaved fabric, nitinol meshes, or high-voltage etched membranes due to opening diameter consistency, constancy of pitch (and hence uniform hole density), and manufacturing ease (i.e., in the difficult to bond dissimilar materials).
Micropore Balloons
|
Lasers can drill many tiny openings in single- or double-walled balloons. |
Balloon angioplasty is a well-known procedure that opens up a coronary artery to treat coronary artery disease. The advent of stents gave cardiologists new options to treat coronary disease, especially with drug-eluting stents that combined pharmacological effects with mechanical scaffolding. This successful mode has carried forth into coronary; peripheral; ear, nose, and throat; heart valve; and spinal treatment as medical designers are combining pharmacology with balloons.
To introduce pharmaceutical agents with an inflated balloon, laser technology is used to drill an array of tiny openings in single- or double-walled balloons. Unlike embolic filters that can be propped open with a nitinol frame, these micropore balloons require openings as small as 5 µm. These openings must be small enough to prevent the collapse or deflation of the balloon, but there must be enough flow to deliver the drug.
During a coronary or peripheral intervention, an alternative to drug-eluting stents is a drug-eluting balloon (DEB), which delivers an antirestenosis drug during balloon inflation. Novel methods to deliver the drug include laser-drilling holes in double-walled balloons. There is considerable interest in using DEBs in the peripheral vascular system. In areas where stents are subject to high torsion and flexion forces (i.e., below-the-knee), stents are subject to fatigue and failure.
Laser drilling 5-µm diameter openings in a 3-D balloon has many technical challenges. Drilling such tiny holes requires imaging with high numerical aperture lenses to achieve the resolution necessary. However, such lenses have an inherently small depth of field (i.e., the image gets out of focus very fast). Nine-axis laser technology with autofocus and mapping capabilities permits these small openings to be drilled on a nonplaner, ever-changing device while keeping the openings round and accurate in size.
The fixturing of the balloons is difficult because ideally, a mandrel should be placed inside the balloon to provide mechanical structure and to maintain balloon shape while the laser drills the holes. Methods using dissolvable mandrels and mandrel-less techniques using proprietary optical systems to limit the depth of field are used. Special techniques are required to prevent the laser from hitting the back wall of the balloon.
In addition to microopening or hole drilling in balloons, lasers can be used to trim the neck of balloons, create microsurface channels, or cut openings.
Catheters and Wires
|
Lasers can change the diameter and taper of holes along the distal length of a catheter. |
|
Laser stripping of polymer coatings of individual wires, bifilars, or catheters exposes underlying conductors. |
Peripheral arterial disease can lead to amputation, especially with patients who have critical limb ischemia. Treatment by adjunctive pharmacomechanical catheter-directed thrombolysis requires devices with precise small openings in multilumen catheters to deliver thrombolytic agents to treat symptomatic, acute, proximal deep vein thrombosi. There are challenges for applying interventional techniques to the superficial femoral artery, popliteal arteries, and distal tibial vessels, and UV lasers can be used for microdrilling, stripping, and skiving operations. For example, drilling holes of typical diameter between 25 and 125 µm with hot needles and mechanical drills are not adequate, because they do not deliver consistent diameters and thus leave jagged or melted edges, and loose chads. Lasers can also dynamically change hole diameter and hole taper, which are important design parameters for engineers concerned with flow rates, especially along the distal length of the catheter.
Laser skiving of catheters such as oval slots and spline cuts are common requirements for a host of applications in coronary, neurovascular, structural heart, heart failure, and other applications that require a catheter-based interventional procedure. Lasers offer the advantage of producing any size or shape with minimal thermal effects on the surrounding material.
Laser drilling and skiving of single or multilumen catheters poses a number of challenges. Multilumen catheters tend to twist longitudinally so the relative position of the laser-drilled holes in the satellite lumens varies. Machine vision in association with laser lathe technology to rotate the catheter is required to compensate for twisting. Preventing the laser beam from hitting the backside of the catheter wall can be accomplished by insertion of mandrels, but it can become problematic if the catheters are too long or the material is opaque, necessitating the use of nonmandrel-based technology to skive catheters.
Atrial fibrillation, cardiac rhythm management, and neuromodulation devices are electromechanical devices. Ablation of the heart to treat arrhythmia (irregular beating of the heart) requires some form of radio-frequency (RF) or electrical stimulation. Laser stripping of polymer coatings of individual wires, bifilars or catheters to expose underlying conductors is a classic laser ablation application. Since the threshold to ablate polymers is an order of magnitude less than the threshold to ablate metals, laser stripping is an ideal selective process. Lasers can remove 100% of the polymer coating. To guarantee a clean underlying conductive surface, the laser beam can overdose the metal surface without any concern of conductor damage. Other methods such as mechanical, chemical, or thermal stripping, leave material residue on the conductor. If the next manufacturing step involves soldering or welding, the joining process may not be reliable.
Selective material removal opens up applications requiring reservoirs, blind holes, and channels that can be applied to a variety of medical products such as balloons, catheters, and wires. Laser ablation has an etch rate of typically 0.1–0.5 µm per pulse. By controlling the number of delivered pulses, the depth can be very precisely controlled.
Bioabsorbable Stents
|
Short-wavelength lasers and those that use short-pulse durations can cut intricate stent patterns. |
Drug-eluting stents, constructed of polymer-coated metal stents, are a significant improvment for treating vascular disease. However, the possibility of late in-stent restenosis and the question of long-term durability and stent fracture introduces opportunities for next-generation stents that are made entirely of bioabsorbable polymers. Lasers of short wavelengths and short-pulse durations, such as excimer, femtosecond, and picosecond lasers, are used to cut the intricate stent pattern fashioned from tubes or flat sheets made of bioabsorbable materials.
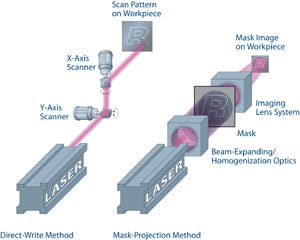
The method of laser cutting stents depends upon the laser source. With excimer lasers, operating at a wavelength of 193 nm, stents are cut by laser-beam illuminating (a noncontact mask) that contains the repeating stent pattern. This image of the mask is then projected onto the target (see the sidebar “Fundamental Principles of Laser Micromachining of Polymers”), providing for a fast parallel process since the excimer laser generates a large beam (typically 25 x 8 mm) and a substantial portion of the stent pattern is cut at the same time. Novel techniques involving the synchronization of laser firing and stages motion can dramatically drive down the cost of manufacture.
Alternatively, lasers that operate at longer wavelengths but have extremely short temporal pulses (femtosecond (~10-15 sec) or picosecond (10-12 sec) can be used to cut the bioabsorbable materials without melting. These lasers cut the stent in a direct-write method in which a focused small spot is traced along the stent pattern. Throughputs can be increased through methods such as beam enhancement and beam splitting, or fast switching (multiplexing).
Unlike the cutting of metal stents, there is no need for the postlaser deburring or electropolishing step of bioabsorbable stents. Eliminating such steps is important, as all machining steps add cost to the device.
Laser choice and laser machining methods are critical to ensure that important mechanical properties of the device (such as radial and tensile strength) and chemical properties (such as surface chemistry) are controlled.
Since bioabsorbable materials are sensitive to temperature and humidity, they are stored in dry nitrogen storage boxes prior to laser cutting. Often inert gases, such as argon or helium, flow in close proximity to the stent during the laser cutting process. After laser cutting, the stents are packaged in hermetically sealed pouches.
Implants
|
Laser micromachining modifies implant surface, promoting more effective adhesion. |
Long-term implants have wide-ranging applications in ocular, orthopedic, and dental. Laser micromachining technology can modify the surface of implants to promote biological integration and increase surface area for better adhesion. By operating a laser at the ablation threshold, periodic sinusoidal structures in metals can be created.
For example, lasers can be used to:
Drill tiny perfusion openings in ocular implants to allow nutrients to pass through the implant and reach the cornea.
Create blind channels direct fluid flow and tissue migration.
Singulate implants without burrs.
Create custom implants—a patient’s specific requirements can be downloaded to a software program and converted to CNC commands for the laser system. Similar to LASIK, which is a customized shaping of a patient’s cornea, the same techniques could be applied to personalizing implants or medical devices in general using higher power lasers, machine vision, and optical techniques.
Conclusion
This article presents a number of exciting medical applications. Reducing healthcare costs requires medical companies to rethink business models to meet the challenges of the global market. The trend towards microfabrication of lower cost polymer-based consumables and devices (both short term and long term) makes UV laser micromachining, using such technologies as excimer, UV-based diode-pumped solid-state laser, femtosecond, and picosecond lasers, an attractive and economically-compelling method to accelerate development and to bring products to market faster. In many cases, laser micromachining is the only viable method, especially as features and tolerances approach the cellular level.
Glenn Ogura is executive vice president of business development at Resonetics LLC (Nashua, NH).
About the Author(s)
You May Also Like