A damage-resistant miniaturized mark can be applied using micromachining to uniquely identify small devices, enhancing tracking capabilities and blocking counterfeiting.
MACHINING
Manufacturers of medical devices and implants are increasingly marking their products. They do so for a number of reasons: to enhance traceability for economic reasons, for long-term quality control, and in anticipation of inevitable future government regulations. Many are also trying to prevent counterfeiting and unregulated distribution. But one- and two-dimensional bar codes have several limitations for some medical device applications. They can be easily read and counterfeited and are usually difficult to miniaturize for placement on small devices. In addition, any defacement can result in information being lost.
One type of coded mark based on encryption theory circumvents these limitations with a marking system that is unique to each manufacturer. These marks could have a large effect on the device industry and its bar coding of devices (see the section “The Future of Bar Codes”). Moreover, this two-dimensional marker identification can withstand significant defacement without information loss. It can also be miniaturized and permanently created on metal and plastic products by use of laser micromachining.
Laser Micromachining Enables Permanent Miniature Marks
Sidebar: |
As with legacy bar codes, there are several ways a two-dimensional marker identification code can be applied to a product or product packaging. These include direct ink printing, printed labels, and surface marking of products. Rigorous implementation of unique device identification (UDI) for medical devices will require direct marking of the product itself. (See the sidebar, “Why Mark Devices and Implants?”) Moreover, the mark must be permanent and inert, which rules out the use of ink in most cases. Therefore, the tool of choice for applying a two-dimensional marker identification is laser marking.
|
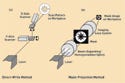
Figure 1. (click to enlarge) Laser micromachining can create marks by using either a direct-write or a mask-based process. Because of its flexibility, the direct-write process is preferable for creating medical device bar codes. |
The two basic methods by which a laser can mark a surface are photomasking and direct writing, as shown in Figure 1. In the photomask method, the laser beam is expanded to fill a mask that often consists of a pattern of reflective chromium on quartz. The pattern created by this mask is then reimaged using lenses onto the surface of the product. This method works well for high-speed applications, but it is only useful if the mark stays the same for each product; any change in the mark requires creating a different mask. Because each identification mark must be different, photomasking will probably not see much use for UDI in the device industry because of its inflexibility.
In the direct-write method, the laser beam is focused to a small spot on the work surface. The spot is rastered across the surface by two mirrors, which are mounted on galvanometer coils to deliver fast computer-controlled angular deflection. The laser power is modulated on and off as the beam is scanned to create virtually any type of mark, from simple alphanumerics to intricate product logos and other patterns. The main benefit of this method, as opposed to a photomask, is its flexibility. The mark can be easily modified in software for each individual product, which makes direct-writing the likely method of choice for most medical devices.
Laser selection is very important. As with other micromachining applications, there are numerous types of lasers, each with its own set of output characteristics and unique advantages. For marking small medical devices and implants, the Q-switched solid-state laser is a good choice. It is often called a diode-pumped solid-state (DPSS) laser, and it produces rapidly pulsed output. DPSS lasers are available with an output wavelength of 1064 nm (near infrared), 532 nm (green), or 355 nm (ultraviolet). The first advantage of this type of laser is that it produces a clean, cylindrically shaped beam that can be focused to a small spot, making it well suited to direct writing. The ability to choose a wavelength also enables users to efficiently mark any material. Most laser manufacturers offer all three wavelengths, and device manufacturers should evaluate a laser manufacturer's process in its applications lab before choosing a particular laser system.
For highly miniaturized marks on metals and plastics, theory and experience indicate that the best results are achieved with an ultraviolet DPSS laser. Infrared and visible lasers micromachine surfaces by acting as an intense local heat source that essentially boils off material. This can result in significant thermal damage in adjacent material, the so-called heat affected zone (HAZ), which can range from an unattractive charred appearance to functional impairment of the product. Moreover, the boiling produces rough edges and recast debris, which may need careful cleaning postmarking. An ultraviolet DPSS laser, however, removes material by breaking interatomic bonds in a relatively cold process called photoablation. This process creates clean edges, and there is almost no recast debris and minimal HAZ. Thermal effects are further minimized by the laser's very short pulse duration (a few tens of a nanosecond). Any peripheral heating that might be created by one pulse dissipates before the next pulse arrives.
Another advantage of an ultraviolet laser is the ability to produce highly miniaturized marks. Specifically, a phenomenon called diffraction determines the smallest spot to which any laser beam can be focused. The size of the spot scales with wavelength, so a laser at 355 nm can be focused to a much smaller spot (e.g., less than 10 µm). For an infrared laser with a wavelength of 1064 nm, a typical spot size can't be much less than 100 µm. Mark miniaturization allows a UDI mark to be created unobtrusively and, importantly, allows marking of tiny devices such as catheters and small surgical screws.
An ultraviolet laser can mark most common device materials, from plastics to tough metals such as titanium alloys. In the case of plastics, a mark can be in the form of a color change or a physically engraved surface mark, depending on the particular plastic and the laser power setting. With metals, the mark can be engraved.
Titanium Medical Implants
Pilot studies are currently under way that examine two-dimensional marker identification of small screws and plates using a marking system that incorporates a solid-state ultraviolet laser. Titanium parts such as screws and plates are used to repair fractures of the mid-face, mandible, and hand, for both neurological and reconstructive purposes. For this reason, screws are generally very small (e.g., 3.2 × 42 mm). In addition, some are as small as 1 mm in diameter with a 1.8-mm-diam head and 2-mm length.
|
Figure 2. (click to enlarge) A UV laser marking system can produce miniaturized two-dimensional marker identification as shown on this titanium screwhead. Photo courtesy of Stryker Micro Implants (Freiburg, Germany). |
These studies demonstrated that tiny screws can be marked with up to 15 bits of information onto a mark that is between 250 and 350 µm square (see Figure 2). These screws and plates are classified as nonsterilized products during transshipments, or shipments that pass through multiple hands (e.g., manufacturers, distributors, hospitals, etc.). They are sterilized at each hospital just prior to use. Hospitals often do not keep records of their use of nonsterilized devices, unlike the case of sterilized products. This can lead to uneconomical use of such products. For example, a distribution center may assemble a manufacturer's products into sets that contain as many 20 individual screws and parts. In many cases, a surgeon will not use all these parts in any given procedure. Without UDI marking, there is no safe way to return these parts to inventory.
The long-term goal is to be able to read the miniaturized marks at every stage of the supply chain using handheld retail-style scanners. This is not a trivial task when the entire mark is only a fraction of a millimeter in total size. Existing conventional scanners don't provide sufficient speed and readability for such small marks. However, there are some prototype scanners designed specifically for this application that are showing promising preliminary results.
Two-dimensional marker identification could allow device companies and their customers to identify counterfeit products. At first glance, devices like screws and plates may seem to be unlikely choices for counterfeiting. However, these are high-value products. Clearly, it is not in the interest of the patient or a device company for unregulated copies to make their way into critical surgeries.
The Future of Bar Codes
In many industries, one of the most commonly accepted methods of applying UDI is to use bar codes that are either linear or two-dimensional. In a conventional linear bar code, the information is coded as alternating dark and light lines of varying thickness and spacing. In a two-dimensional bar code, the information is coded in an array of dark and light square pixels.
|
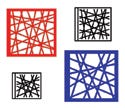
Figure 3. (click to enlarge) The two-dimensional marking identification system consists of multiple intersecting straight lines within a square or rectangle. |
In a two-dimensional marker identification code, a computer algorithm converts the binary or alphanumeric information into a rectangular or square pattern of intersecting straight lines that the laser will cut into a device. Typical examples are shown in Figure 3. The position, angle, and thickness of these lines determine the coded information that is carried.
|
Figure 4. (click to enlarge) Two-dimensional marker identification can be significantly defaced, as in this example, without losing any information content. |
There are several significant advantages to the two-dimensional marker identification approach. First, it is relatively immune to information loss through damage. For example, if part of a traditional bar code is obliterated, then that segment of the coded information is permanently lost. But with two-dimensional marker identification, as long as the reader can scan some part of every line, then the system software can readily reconstruct the entire image and thereby decode all the original information. In fact, an entire corner or most of a side of a two-dimensional marker identification mark can be obliterated without any loss of information (see Figure 4). This is particularly useful in multiple-use and high-wear products.
But for medical device manufacturers, a more important advantage of two-dimensional marker identification is that it enables proprietary coding. The coding of conventional bar codes is standardized, so they can be read and duplicated by anyone to re-create them on counterfeit products. With two-dimensional marker identification, complex proprietary encryption algorithms generate and decode the marks. Moreover, these algorithms support almost infinite combinations of coding.
There is also the issue of miniaturization, which can be very important for manufacturers of small devices and implants. There is a limit to how small conventional bar codes can be generated and read. The pixels and lines are close together and need to maintain certain sizes. But a laser micromachined two-dimensional marker identification is a relatively open mark with well-spaced lines. Therefore, it does not require the resolution or contrast of a conventional mark. This allows a two-dimensional marker identification mark to be used in a much smaller format; the mark can be a fraction of a millimeter in size and thus can be completely unobtrusive. In addition, high-quality lasers are capable of machining very small marks onto the devices.
Conclusion
The benefits of having a coded marking system are significant. Advances in laser micromachining have made such systems possible, particularly for small devices that need small bar codes. Lasers used today can machine such marks without being plagued by thermal damage and debris concerns. In addition, lasers that leverage low-heat techniques can be used to machine plastics as well as metals. As the medical device industry faces the possibility of mandated UDI, these types of machining systems will play an important role in the future of medical devices.
Acknowledgment
The authors would like to thank Dieter Franki, director of Stryker Micro Implants and Resorbable Solutions (Freiburg, Germany) for his input and assistance.
Sri Venkat is director of marketing for Coherent Inc. He can be reached at [email protected]. Kevin T. Simmons is vice president and managing director of global sales for ORBID ( San Francisco). His e-mail is [email protected].
Copyright ©2007 Medical Device & Diagnostic Industry
About the Author(s)
You May Also Like