Tweaking a supplier’s valve resulted in enhanced comfort for prosthetics patients
May 1, 2008
Originally Published MPMN May 2008
ENGINEERING EXCELLENCE
Infant Warmers Feature Improved Usability and Safety Functions
|
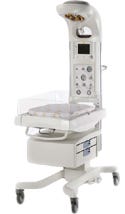
The redesign of the Panda iRes and Giraffe infant warmers included adding more safety features and improving the units' maneuverability. |
Previous models of infant warmers served their purpose. Infant warmers from GE Healthcare (Laurel, MD; www.gehealthcare.com) have been redesigned to serve people—the babies they protect and the healthcare providers who control the units. After five years of researching customer, patient, and clinical needs, GE has loaded the Panda iRes
and Giraffe infant warmers with new safety features, improved the units’ ergonomics and maneuverability, and simplified control panels.
One aspect of the redesign involved integrating respiratory support into the units to comply with changes in infant resuscitation guidelines from the American Academy of Pediatrics’ Neonatal Resuscitation Program. “GE designed its new resuscitation system for the warmers with these guidelines in mind,” says David Ball, general manager, infant care, at GE. “The system integrates blended gas, T-piece resuscitation, and SpO2 monitoring; requires minimal setup time; and helps standardize resuscitation protocols across the perinatal care area.”
The manufacturer brought in a partner, Biomed Devices Inc. (Guilford, CT; www.biomeddevices.com), to help design the new resuscitation system and integrate the gas components. The most challenging aspect was combining the gas-delivery system, the suction system, and the air-oxygen blender into one package that could then be inserted into the tower of the infant warmer. Biomed also helped GE devise a safety override system for the positive inspiratory pressure (PIP) function. “The PIP override allows the therapist to limit the pressure to normal operating pressures but, in extreme cases, they can temporarily override it, depending on the patient’s needs,” says Nolan Holabird, production and engineering manager for Biomed.
Other new features also increase the amount of control that healthcare providers have over the units. A hands-free alarm silencer enables clinicians to turn off alarms by simply waving a hand in front of a sensor so they can stay gloved while attending to infant patients. The heater heads of the units are recessed to eliminate overhead obstacles and to improve access to the infant while also providing uniform heat across the unit’s bed.
Recognizing the need to incorporate even more aesthetic and ergonomic improvements to the redesign of one of its flagship product lines, GE turned to HumanCentric Technologies Inc. (Cary, NC; www.humancentrictech.com). “HumanCentric helped us maintain a family-friendly design that could transition to a critical-care environment,” Ball says.
Integrating human factors concerns with ergonomic and industrial design elements is HumanCentric’s specialty. What was difficult for the design firm was figuring out how to make an approximately 500-lb unit easy to maneuver for a typical nurse. “The real challenge was not only developing something that would be easy to push, but also developing it in such a way that the handles and the points where force would be exerted would be properly and ergonomically located to maximize the force generated and reduce, if not eliminate, the potential for back problems,” says Barry Beith, HumanCentric’s president.
The cart of the infant warming unit has to be stable enough to carry gaseous bottles, like oxygen bottles, and is weighed down by batteries used to run the unit while in transit. Beith notes that lighter-weight battery power, if it ever becomes available, could improve the device—but the technology isn’t there just yet. The company instead focused on the other areas it could improve.
To help eliminate staff fatigue from bending over infant warmers, GE and HumanCentric raised the height of the infant bed. The neck of the Giraffe unit was reconfigured so that the light could be more easily controlled by the nurse, but also would not be so close that it would create hot-spots or discomfort for the infant. Unlike earlier models, the new warmers feature a lip around the weighing mechanism for added patient safety.
And to further protect the infants, HumanCentric was able to simplify the control panel using multicontrol functions to reduce the number of displays and the potential for human error. “The project was absolutely fascinating, not only with regard to redesigning something almost completely, but also producing a workstation that was better for the patient [and] more readily usable and functional for the staff in the birthing centers,” Beith says.
“Human factors aspects of design in medical products are becoming ever more critical to reducing and managing human error, to our ability to move safe equipment from institutions into homes, and to our ability to make our designs work better,” Beith adds.
Copyright ©2008 Medical Product Manufacturing News
You May Also Like