May 1, 2008
|
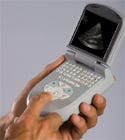
The Acuson P10 is the world's first pocket ultrasound. |
In the next phase of ultrasound evolution, it may be commonplace for clinicians to tote around a handheld ultrasound unit during their rounds and use it as part of a standard physical. This capability could vastly improve point-of-care diagnostics by providing immediate proof of what physicians may suspect they hear or feel.
Touted by its OEM as the world's first pocket ultrasound, this next-generation imaging tool has earned a Medical Design Excellence Award and comes courtesy of Siemens Medical Solutions (Mountain View, CA; www.siemens.com). Already on the market, the Acuson P10 handheld diagnostic ultrasound unit is for 'a quick look right here, right now' use, according to Michael Elser, P10 product manager. "It's really designed to be a personal imaging tool, bringing ultrasound closer to point-of-care assessment anywhere immediate visual information is going to help that clinician decide how to care for a patient," he says. Elser points to cardiology, OB/GYN, the ICU, and the emergency room as areas that could benefit from the product.
Siemens was aware that the approach to developing a handheld ultrasound simply could not mirror that for a traditional unit. The challenge was in determining which features were necessary. "If you think about trying to squeeze all of that traditional functionality into as small a package as possible, there are many trade-offs as to what functionality is in and what is not," Elser says.
More than a year of research revealed that a potential P10 user's needs ultimately differed from those of Siemens' typical ultrasound end-users in the diagnostic and therapeutic realms. "We're bringing them much further up into the discovery phase of the health journey, so you look at who might be using this device," says Stephen Hooper, president of Elemental8 Inc. (San Jose; www.elementaleight.com), a firm formerly owned by Siemens that was charged with the product's industrial design. "You now are looking at what is the most appropriate solution for these new users." With this product, the definition of a typical end-user would likely reach beyond an ultrasound technician to include physicians, nurses, and EMTs.
The key to the design was targeted functionality. "What is typical in the market is to create feature-rich solutions and the trend is now moving toward feature-specific products," Hooper says. Overloading the device with features would also drive up the cost, which was understandably intended to be lower than that of a traditional ultrasound.
To produce the complex unit that research showed users wanted, Siemens required the additional expertise of Acorn Product Development (Fremont, CA; www.acornpd.com), which helmed the mechanical design of the P10, and GMT Design Services (Boulder, CO), whose system architect, Gregg Miller, served as the project's system engineer and was integral to the architectural work.
"There were many iterations to come up with an overall concept that met these competing needs: [having] enough volume to fit the electronics, the battery, and the display, and [a product that] was comfortable in your hand and mechanically viable from a manufacturability, reliability, and cost perspective," Elser says. "Those firms, working along with internal experts, were key in driving that evolution to closure."
Among the hurdles that the team had to clear were thermal engineering issues related to the amount of power pumped through the system. As a handheld device, the P10's threshold for surface contact temperature is low and it was necessary to develop a method for dispersing the heat throughout the system, according to Robert Jetter, director of mechanical engineering at Acorn. Jetter adds that EMI proved to be another major challenge, which the team addressed by using "every trick in the book" to reduce the amount of noise emitted by the device.
With the finished product, Elser believes that the potential is limitless. "My opinion is that, over time, any practitioner that physically touches a patient benefits from this technology."
Copyright ©2008 Medical Product Manufacturing News
You May Also Like