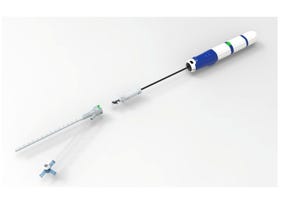
Obsidio Conformable Embolic
Regulatory & Quality
Boston Scientific Class I Recall Linked to Two DeathsBoston Scientific Class I Recall Linked to Two Deaths
Categorized as a correction, not product removal, the company is recommending physicians not use the aliquot technique to deliver the Obsidio Conformable Embolic due to increased patient risk.
Sign up for the QMED & MD+DI Daily newsletter.



















.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)


.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)

.png?width=300&auto=webp&quality=80&disable=upscale)
.png?width=300&auto=webp&quality=80&disable=upscale)



.png?width=300&auto=webp&quality=80&disable=upscale)