Originally Published MDDI April 2006MDEA 2006 Niche markets provided these winning manufacturers with opportunities to develop life-improving devices for small or undertreated patient populations. Erik Swain
April 1, 2006
MDEA 2006
It seems that only a limited number of medical device types—usually those that have big effects on lucrative markets—grab the headlines. In reality, however, those devices that draw media attention are the exceptions to the device development process, not the rule. The vast majority of devices are sold to niche markets.
But developments in those niche devices shouldn't be underplayed. They often represent major quality-of-life improvements to small or neglected patient populations. And they can improve ease-of-use for caregivers to these patients. Nursing-home aides and ambulance workers may not have as much clout as high-profile cardiac surgeons, but they save lives, too.
The products highlighted in this article won Medical Design Excellence Awards because they represent significant advancements for underserved markets and underappreciated patient and caregiver populations. Their benefits may not be as obvious as those of some other winners, but their contributions to healthcare are no less important.
|
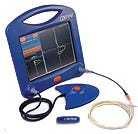
The Cortrak system's electromagnetic sensing device tracks the path of feeding tubes during insertion to ensure proper placement. |
The Cortrak System
Feeding-tube placement is a routine but crucial task. Many times, though, it is performed blindly. If the tube is mistakenly lodged in the lung, it can cause aspiration or other pain and discomfort for a patient. Moreover, it necessitates using an x-ray to verify the location of the misplaced tube, exposing patients to extra radiation.
Viasys Healthcare/Medsystems (Wheeling, IL) developed the Cortrak system to improve the feeding-tube placement process and reduce x-ray use. “There has been a need to lessen patient exposure to x-rays during feeding tube placement,” says George Nassif, marketing services manager of Viasys. “We had talked with customers about the difficulty of getting tubes into the small bowel. It often required multiple x-rays to ensure the feeding tube was in the right place. So we decided to apply engineering principles to develop a new system.”
The Cortrak uses an electromagnetic sensing device to track the path of feeding tubes during placement. It is usually used in conjunction with an x-ray, but the x-ray should not be needed more than once. It won an award in the General Hospital Devices and Therapeutic Products category.
The device consists of a monitor unit, a receiver unit, and a transmitting stylet. The stylet, which has a coil winding at its end, is placed within the tube and transmits a signal. The receiver tracks the signal, and the monitor processes and displays it.
One key to the design was making the phase detection circuit very sensitive. This allows the system to pick up even very weak signals. It also enabled the signal to be of very low amplitude. This was important because it would minimize interference with other electronic equipment and allow the current feed to be very low, causing no harm to the patient in the event of current leakage. The design also enables power to be cut in milliseconds should circuit integrity be compromised.
The interface was designed with ease-of-use in mind. Navigation and data entry are done via a touch screen. And the system is portable, making it valuable to a multibed facility like a nursing home.
“This product has the potential to improve patient safety by eliminating the adverse effects associated with misplaced feeding tubes,” says juror Jay Goldberg, director of the Healthcare Technologies Management Program at Marquette University (Milwaukee). “It will also help reduce costs associated with the secondary placement resulting from misplaced tubes.”
Indeed, although Viasys projects savings at $25,000 per year, some jurors thought that was a low estimate.
“If anything, I believe that they were somewhat conservative in their estimates because they did not incorporate any cost differential for a patient who ended up with aspiration pneumonia,” says juror Michael P. Schollmeyer. “I thought it was a very worthwhile and practical product.” Schollmeyer is director of clinical research at CHF Solutions Inc. (Brooklyn Park, MN).
The tracking technology could also eventually be adapted to other applications, says Nassif.
Denlite Illuminated Dental Mirror DP5000
You might not think the dental mirror needed improvement—but then, you probably haven't talked to dentists about it. Welch Allyn Inc. (Skaneateles Falls, NY) did, and the company used their input to design a more-useful and less-cumbersome device, the Denlite Illuminated Dental Mirror DP5000.
|
The Denlite Illuminated Dental Mirror is cordless, which prevents failure modes associated with corded dental mirrors. It also reduces exam times. |
“This is one of those simple, ‘that's a great idea' devices,” says juror Mary Beth Privitera. “The light is extremely bright and the clarity of the mirror is exceptional.” Privitera is assistant professor of biomedical engineering for the University of Cincinnati's Medical Device Innovation & Entrepreneurship Program. The Denlite won an award in the Dental Instruments, Equipment, and Supplies category.
The Denlite is not the first illuminated dental mirror, but it is the first cordless one. “The previous product was corded, and it would snag in the dental chair,” says Richard A. Tamburrino, Welch Allyn's engineering manager. “The number-one failure mode was the cord.” So the Denlite runs on batteries that can be recharged in less than 15 minutes.
The company took the opportunity to make other improvements as well. The previous model had a halogen bulb that would get hot. If a dentist held it long enough, he or she could feel heat on the handle, which could be hazardous for the dentist and could lead to errors affecting the patient. So Welch Allyn replaced it with an LED light cartridge, which does not get hot. Also a plus, its white light provides brighter illumination than halogen's yellow light. It's so bright, in fact, that overhead light is not needed. Since the dentist doesn't have to adjust overhead lights or power cords, the Denlite cuts down exam times.
The company improved the mirror, too. Welch Allyn collaborated with Alliance Precision Plastics (Rochester, NY) to develop an automated in-mold decoration process to produce one-piece mirrors. They were made from K-Resin and a proprietary metal foil. (In-mold decoration had never before been used to make a medical product.) The design enables the mirror to transmit light efficiently and remain strong enough for cheek retraction and tongue depression. It is also compact enough for patient comfort.
The handle was designed to be robust enough to endure thousands of steam sterilizations and flat enough that it wouldn't roll off a tray.
Being cordless, the instrument is portable, so it can be taken outside the dentist's office if necessary.
“This is a very clever design that overcame some technical challenges to bring it to fruition. And it appears to be highly functional,” says juror William Hyman. “A seemingly simple device can be an award winner if it is well designed and well presented.” Hyman is a professor at Texas A&M University's department of biomedical engineering, located in College Station, TX.
|
The Impact hearing-aid battery recharger works on sunlight alone and can recharge almost any type of hearing-aid battery. Project Impact and industrial design firm Bridge Design worked to make the recharger look like a consumer electronics device. |
Impact Solar-Powered Hearing Aid Recharger
In the United States, a hearing aid costs about $1500 per ear. That would seem unaffordable to residents of third-world countries, but that didn't stop the Impact Foundation, a Berkeley, CA–based nonprofit organization that runs a program called Project Impact.
“Poverty and disability go hand in hand,” says Joel Segre, a project manager for Project Impact who came up with a low-cost hearing aid idea while in college. “The more impoverished one is, the harder it is to get medical care; and the harder it is to stay healthy, the harder it is to be able to work. A hearing aid can help break that cycle.”
The organization developed a hearing aid that costs only $50 per ear, and it made them available for little to no cost to those in need. But one obstacle remained: battery technology. Hearing-aid batteries are not cheap, but the recipients of the device may be earning as little as $1 per day. So Project Impact developed a recharger that works on sunlight alone and can recharge almost any type of battery for almost any type of hearing aid.
“It had to be able to withstand extreme heat and extreme rain, so we used UV-absorbable resins,” Segre says.
The initial prototype was rudimentary, since costs had to be kept down. “Our feeling was that the people who would use this would care about price and functionality but not cosmetics,” says Segre, who tested the prototype in India. “That was forcefully disproved. People rejected it, saying it looked like it was low quality.”
So Project Impact hooked up with industrial design firm Bridge Design (San Francisco), which agreed to do most of its design work for free. It reduced the number of parts and made the product look like a consumer electronics device, which is a desirable quality even in the poorest communities. It has just three parts, which combine the functions of case, battery door, battery holder, solar panel, and charging contacts.
“The obstacles of the initial prototype were that people didn't like it and retailers didn't want it shown in their showrooms,” says Segre. “The new instrument can be worn and displayed with pride.”
The recharger is now available in India, Pakistan, Nepal, Sri Lanka, and East Africa. The World Health Organization has expressed interest and may get involved with distribution. That would expand its availability exponentially.
Jurors were impressed with the technology's ability to provide care to the too-often uncared-for. They gave it an award in the Rehabilitation and Assistive Technology Products category.
“For emerging countries, the ability to recharge hearing-aid batteries is an extremely innovative idea to bring people with fewer resources the gift of hearing,” says Tor Alden, a principal at HS Design Inc. (Gladstone, NJ).
“This device brings healthcare to those who are less fortunate and have no alternatives,” says Privitera. “It provides an example of leadership to develop devices with severe cost constraints and supports an environmentally friendly technology. Kudos to those parties involved to make this project a reality and for demonstrating successful development of an ecologically sensitive, accessible device.”
Rugged Power Pro XT Ambulance Cot
There are many ways healthcare workers get injured on the job. One that does not receive much publicity is emergency workers who hurt their backs from lifting cots carrying patients. This has become a more serious problem in recent years with the rise in obesity in the United States. There's already a shortage of paramedics, and early retirements due to back injuries is making the situation worse.
|
The Rugged Power Pro XT Ambulance Cot is lightweight and battery powered, which helps prevent back injuries in paramedics caused by lifting overweight patients. |
In response, Stryker EMS (Portage, MI) developed the first hydraulic cot for the U.S. market. It received an award in the Critical Care and Emergency Medicine Products category.
“This has been a crying need for the past three to five years,” says Jason Kneen, a design engineer at Stryker EMS. “As patients get bigger, operators' backs have had to take on more and more weight. We wanted to design something that would allow paramedics to not have to lift a cot.”
The company had to design a powered lifting device that was lightweight, dual speed, and battery powered. The result, the Rugged Power Pro XT, comes with full manual backup and built-in safety features for both the powered and the manual modes. It was subjected to extreme testing to verify its ability to withstand intense heat, cold, humidity, vibration, and sudden impact.
And the cot's electronics and hydraulics were so robustly designed that it was the first product of its kind to receive certification in IEC 60601 compliance from Underwriters Laboratories. “That guarantees to the medical community that the cot will not interfere with heart monitors or any other devices,” Kneen says.
Of course, getting certification meant overcoming a number of engineering challenges. The cot had to support patients up to 700 lb while weighing less than 130 lb itself. So its base uses dual telescoping X-frame leg technology with a four-bar design to reduce the loads on the base geometry. It had to have sensing technology to know when to accelerate or decelerate the speed of motion. It needed two speeds: fast for grave emergency cases, and slow for less urgent cases to provide better patient comfort. And it needed push-button retraction for when the patient was loaded into the ambulance. (Manual loading takes 10–15 seconds, whereas an automated load can be done in as little as 2.5 seconds.)
The response has been overwhelming, says Kneen. “We met all our financial goals for a year within the first three months after launch,” he says. “The demand is bigger than what we can put out, and that says a lot.”
“This is an excellent product,” says juror Denise M. Korniewicz. “It reduces back injury among EMTs and provides safety for heavyset patients. Patients do not fall off the cot, and they are not uncomfortable and jolted around because of a small cot.” She is professor and senior associate dean for research at the University of Miami (FL) School of Nursing.
|
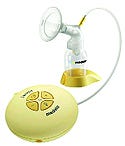
The Swing Breast Pump promotes milk ejection and simulates a baby's nursing rhythm, delivering maximum milk flow in a short time. |
Swing Breast Pump
In today's hurried society, breast pumps are becoming more of a necessity. This may not be a subject that's discussed in public much, but that doesn't preclude customers from demanding better designs. With its Swing breast pump, Medela Inc. (McHenry, IL) met this demand.
“We needed a single electric pump that met moms' criteria: small, lightweight, battery operated with enough battery life, stylish, and quiet,” says Terry McCloud, Medela's marketing communications manager. Also, he says, there had to be a better method for suction.
Medela developed the first two-phase pumping program for home-use breast pumps. Previously, the technology was found only in hospital pumps. The two phases are stimulation and expression. The former promotes the milk-ejection reflex. The latter simulates a baby's actual nursing rhythm and can deliver maximum milk flow in less time.
The challenge was to develop a small pump that had the power of its bigger competitors without hampering product durability and lifespan. It also had to be lightweight, mobile, and easy to clean. Medela's design is versatile enough to be used on a tabletop, worn around the neck or over the shoulder, or clipped onto a belt.
The initial design was tested by 150 mothers in eight countries. “We received very positive feedback on the product on numerous fields of measure, even from moms who experienced some problems with the initial design,” says McCloud. “After some design modifications, our second field testing, conducted with 80 moms in four countries, garnered the same high marks with better performance in the improved areas. Most moms wanted to keep the product indefinitely because it performed and was designed far better than other single electric pumps they had used. We also received very positive feedback on the look and functionality of the product. Technical and product testing exceeded all performance goals set for the product.” Of particular help was an easy-to-use control panel that allows mothers to make manual adjustments in midpump if they need. The pump also can be used with one hand.
“The designers of this product understood the needs of the potential users of this device and provided features to meet those needs,” says Goldberg.
The pump won an award in the Over-the-Counter and Self-Care Products category. It launched in the European market in the fall of 2005 and is expected to be available in the United States in the summer of 2006. Medela expects to use the two-phase pumping technology on other products.
“This is a user-friendly consumer device that was well thought out,” says juror Gail D. Baura, vice president for research and chief scientist at CardioDynamics (San Diego).
Copyright ©2006 Medical Device & Diagnostic Industry
About the Author(s)
You May Also Like