October 1, 2002
Originally Published MPMN October 2002
INDUSTRY NEWS
Antimicrobial Wound Dressing Introduced
Kelly Donoghue
|
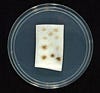
A medical foam for wound dressings is shown with and without an antimicrobial additive developed by Milliken Chemical. |
A silver-ion material that prevents bacterial contamination has been used as an additive in a medical foam for wound dressings. Developed by Lendell Manufacturing Inc. (LMI; St. Charles, MI; www.lendell.com), Microbisan is the first absorbent dressing to contain antimicrobial Alphasan from Milliken Chemical (Spartanburg, SC; www.milliken.com).
LMI and Milliken jointly developed the product, according to Phil Sheridan, LMI technical and research director. The antimicrobial is added to the foam by means of a polyurethane production process that completely disperses the additive throughout the finished product. The antimicrobial in the foam keeps the wound free of bacterial contamination as the dressing absorbs wound fluids. Alphasan is effective over long periods, continuing to work even when dressings are infrequently changed.
A zirconium phosphate–based ceramic ion-exchange resin containing silver, Alphasan is effective against a range of microorganisms that can cause odor, discoloration, biofilm formation, and other undesirable effects. The material is widely used in Europe, Japan, and the United States, and it is approved by FDA for contact applications.
Copyright ©2002 Medical Product Manufacturing News
You May Also Like