3D Printing
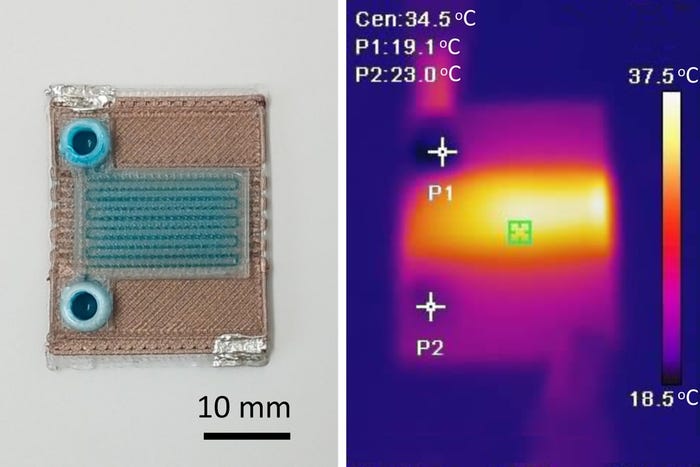
3D-printed implantable lower-limb socket
3D Printing
New Bodies of Work in 3D PrintingNew Bodies of Work in 3D Printing
3D printing is taking the manual labor and guesswork out of reconstructive and implantable device design while improving patient comfort.
Sign up for the QMED & MD+DI Daily newsletter.

.png?width=700&auto=webp&quality=80&disable=upscale)