October 29, 2013
While countless tubes are extruded or injection molded each year, these manufacturing techniques are neither the most optimal nor the most cost-effective methods available to medical device manufacturers. At least that's the word from Avalon Laboratories (Rancho Dominguez, CA), whose polymer solution casting technology can be used to manufacture flexible plastic components without resorting to conventional extrusion or injection-molding processes.
|
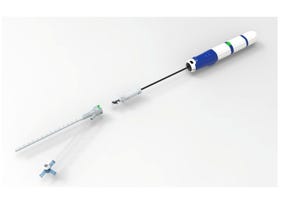
A coil-reinforced catheter with introducer is manufactured using Avalon Laboratories' polymer solution casting technology. |
Polymer solution casting begins with a mandrel or an inner-diameter mold that is carefully immersed into a tank of tightly controlled polymer solution, explains Michael Janish, Avalon Laboratories' president and CEO. Responding to a combination of thermal and frictional properties, the liquid plastic forms a thin layer around the mold, which is then extracted from the bath and cured. Once the first layer of the thin film has solidified adequately, other features can be added to the product, such as coiling, braiding, other types of metal reinforcements, and even imaging targets. Multiple casting steps can then be repeated to build up wall thicknesses, add additional lumens, and optimize column strength. After it is totally cured and solidified, the part is removed from the mold.
This casting technology, Janish says, was developed for the manufacture of cannulae, a subset of catheters that is typically used for directing blood flow. While it can be used for many other applications, the method is commonly used to manufacture cannulae used in open-chest procedures to provide extracorporeal membrane oxygenation.
"This thin-film processing technology is unique because it can meld materials with different properties--including strength, durometer, color, and lubricity," Janish remarks. "For example, the first layer can be made out of a lubricious material to enable the passage of such medical devices as stents, while the outer layer can be composed of material that adds column strength. Alternatively, a layer can be made from materials offering a range of different cytotoxicity properties or lubricious characteristics so that a physician can push the tube against an artery or a vessel tissue in the body."
In high-mix, low-volume applications, this technology frequently costs less than more-automated extrusion or injection-molding processes because it relies on the use of simple tooling, according to Janish. "Our mandrels and molds in most cases are axisymmetrical, meaning that they're made on a lathe and a grinder. While complicated extrusion die heads or tooling for such applications as inner lumens require the use of a mandrel in between the tooling, our process can create multiple lumens using piano wires and other axisymmetrical molds to create the additional lumens.
In typical extrusion processes, operators pour pellets into one end of a machine to melt and blend them into a gel. Then, the material is pushed through a die head, from which it emerges in the shape of tubing. "The problem with this method is that you usually can't perform many alterations," Janish notes. "The tubing is consistent from the distal to the proximal end--from the tip to the tail. And while you can vary the speed of the machine a little bit and perform bump extrusion to modify the inner or outer diameter, you can't vary the features of the tube very much." To form features, the tubing must be run through another machine to add such reinforcements as braiding or coiling, Janish adds. And in most cases, you have to take a piece of another extruded tube, place it over the top of the first tube, and then shrink the assembly together using a heat gun to create the final construction.
In the catheter-manufacturing space, the inner-diameter surface of a tube is often composed of a different material from the outer diameter surface. For example, because doctors are used to feeling their way through a catheter as they press a stent or other device through it, they frequently prefer that the inner liner be composed of PTFE. Avalon's technology enables users to mount an extruded piece of PTFE or other material on a mandrel and then cast polymer on top of it. This process, Janish says, enables manufacturers to embed extruded components in their tubing. "If a multilumen part containing a polyimide lumen is needed to achieve such attributes as column strength, high pressure, or consistency at body temperature, we can embed a polyimide-extruded tube inside of our solution casting technology."
About the Author(s)
You May Also Like